Product Images Azithromycin Dihydrate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Azithromycin Dihydrate NDC 68180-861 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image - 1ede87c9 811c 4887 a274 a248a1e58a9d 02
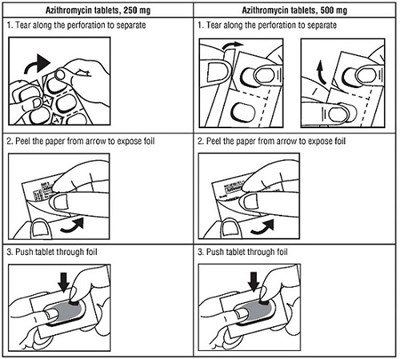
This is a label/instruction for Azithromycin tablets of different strengths - 250 mg and 500 mg. There are instructions to tear along a perforation to separate, peel off paper from an arrow to expose the foil, and push the tablet through the foil. This appears to be part of the medication packaging.*
Bottle Label - 30 Tablets - 250 mg - 1ede87c9 811c 4887 a274 a248a1e58a9d 03

This is a medication called Azithromycin manufactured by Lupin Limited. Its visual dosage can be found in the accompanying prescribing information. It can be taken with or without food and should be stored at room temperature. The medication comes in the form of tablets, and it should be dispensed in a tight container as defined by USP. Pharmacists should provide the medication along with a patient information leaflet.*
250 mg - 1ede87c9 811c 4887 a274 a248a1e58a9d 05
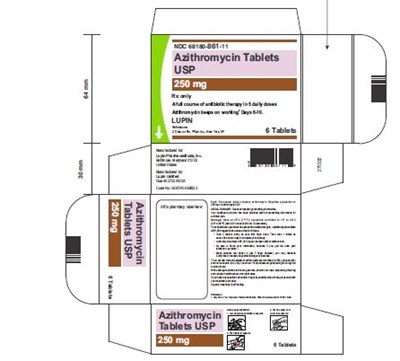
Azithromycin Tablets USP is a type of medication used to treat bacterial infections. It belongs to the class of macrolide antibiotics and is commonly prescribed for respiratory tract infections, skin infections, and sexually transmitted infections. The tablets are taken orally and work by preventing the growth and spread of bacteria in the body.*
Bottle Label - 30 tablets - 500 mg - 1ede87c9 811c 4887 a274 a248a1e58a9d 07

Each film-coated tablet contains azithromycin dihydrate equivalent to USP. The usual dosage can be found in the accompanying prescribing information. Azithrol Mycin can be taken without food. The tablets should be stored at 25°C (77°F) with excursions permitted to 15° t030°C (59° to 86°F). Lupin Limited is the manufacturer and the tablets should be dispensed in tight containers as defined by the USP. This is a prescription-only medication and should be dispensed with the patient information leaflet provided separately to each patient.*
500 mg - 1ede87c9 811c 4887 a274 a248a1e58a9d 09
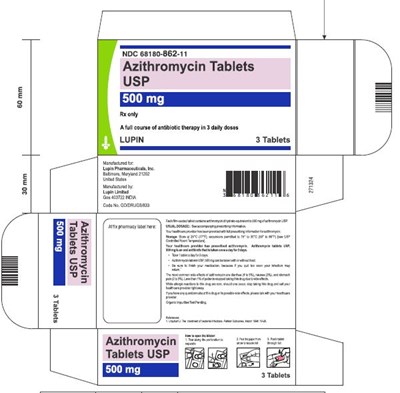
This is a description of Azithromycin Tablets USP with the NDC code 68180-862-11. The remaining text is not readable or relevant.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




