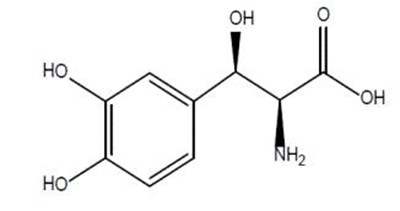
Product Images Droxidopa
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Droxidopa NDC 68180-987 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image-02 - 73d75f21 7817 4a1a 9016 168526fa1c71 02
This text does not provide enough context or information to generate a useful description.*
image-03 - 73d75f21 7817 4a1a 9016 168526fa1c71 03

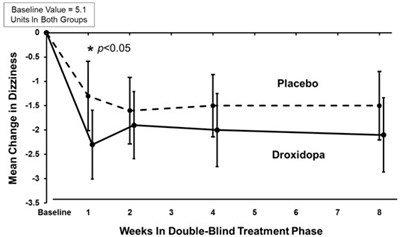
This appears to be a chart or graph displaying the improvement in OHSA Question 1 for subjects receiving a placebo or droxidopa, with a scale on the left side indicating the percent of subjects and a scale on the bottom indicating the rating of improvement. The chart shows that a higher percentage of subjects receiving droxidopa reported improvement compared to those receiving placebo.*
Image - 73d75f21 7817 4a1a 9016 168526fa1c71 04

This is a description of a medication packaging of droxidopa capsules manufactured by Lupin Pharmaceuticals Inc. Each capsule contains 100 mg of droxidopa. Specific instructions on dosage can be found in the prescribing information. The medication should be stored at room temperature in a tightly sealed, light-resistant container, away from the reach of children. The packaging contains 90 capsules, with a printed seal under the cap that should not be broken or missing before use.*
Image - 73d75f21 7817 4a1a 9016 168526fa1c71 05

This is a prescription medicine from Lupin pharmaceuticals with an NDC number 68180-988-09. It is Droxidopa capsules and each capsule contains 200 mg of droxidopa. The usual dosage is specified in the prescription information. It shouldn't be used if the printed seal under the cap is broken or missing. Its storage temperature should be between 20°C to 25°C (68°F to 77°F); with the permitted excursion of 15°C to 30°C (59°F to 86°F) as mentioned in the USP controlled room temperature. Only dispense it in a closed, light-resistant container with a child-resistant closure. The medicine is manufactured by Lupin Limited in Nagpur, India, and distributed by Lupin Pharmaceuticals, Inc. from Baltimore, Maryland, US. The warning advises keeping it out of the reach of children. Code no. MH/DRUGS/25-ND/59, 266612 is mentioned on the packaging.*
Image - 73d75f21 7817 4a1a 9016 168526fa1c71 06

This text provides information about a medication with the National Drug Code (NDC) 68180-989-09, which contains 300 mg of droxidopa per capsule. The usual dosage instructions are not provided and must be obtained from the prescribing information. The manufacturer is Lupin Pharmaceuticals Inc., with a warning not to use if the printed seal is broken or missing. Storage instructions include storing at a temperature range of 20°C to 25°C (68°F to 77°F) with limited excursions, and at a permitted range of 15°C to 30°C (59°F to 86°F). Dispensing should occur in a tightly sealed, light-resistant container with a child-resistant closure. A warning to keep the medication out of the reach of children is included. The raw text includes some errors and typos.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.