Product Images Alendronate Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Alendronate Sodium NDC 68258-3014 by Dispensing Solutions Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label Image for 70mg 4 Count Bottle - NDC 68258 3014 01

The text describes a bulk source data for a product manufactured by Cobalt Laboratories, with a product ID of a white to off-white oval tablet, embossed with AN70/ company logo. The bulk source NDC is 16252-0601-44 and each tablet contains 91.37mg of alendronate sodium USP (70mg free acid equivalent). The product is enclosed in a tight or light-resistant container, and it should be dispensed as per the package instructions unless otherwise directed by a physician. The text specifies that the medicine should not be taken while lying down, eating, drinking or while taking other medications for at least 30 minutes. The text provides the contact details of DispenseQuick, the company that packaged the medicine, and warns that it should be kept out of children's reach and stored between 68°-77°F. The description also provides a lot number (5A0244), an MN number (16252060144), and an RX number (20660941).*
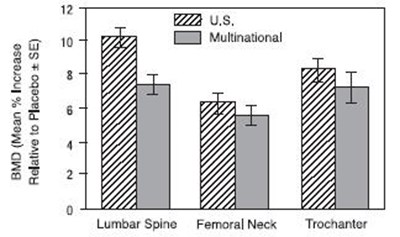
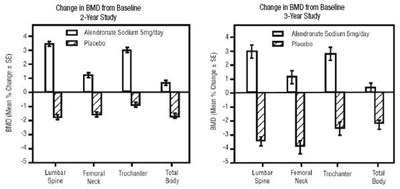
figure - figure1and2

This appears to be a table containing data for a study involving a drug called "Alendronate Sodium" at a concentration of 10mg. There is also a control group receiving a placebo. The data appears to be organized by month, with data points at 0, 12, 24, and 36 months. There are some numbers in the table that are not clear due to errors.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.