Product Images Levetiracetam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Levetiracetam NDC 68258-7089 by Dispensing Solutions, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
NDC 68258-7089-XX - NDC 68258 7089 XX AUROBINDO
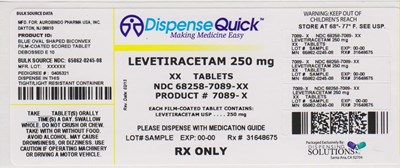
This is a description of a medication produced for Aurobindo Pharma USA, INC. in Dayton, NJ. It is a blue oval shaped biconvex scored tablet with E 10 debossed on it. The bulk source NDC is 65862-0245-08, and the mfr. lot is XXXXXX. The medication is Levitiracetam 250mg, and there are XX tablets in one package with product number 7089-X. The medication should be stored between 68°- 77° F, and kept away from children. It may cause drowsiness or dizziness, so caution should be used when operating machinery or driving. There are instructions to take the tablets orally, whole, and not to chew them, or crush them. They can be taken with or without food, and should be dispensed with a medication guide.*
Figure 1 - levetiracetam fig1
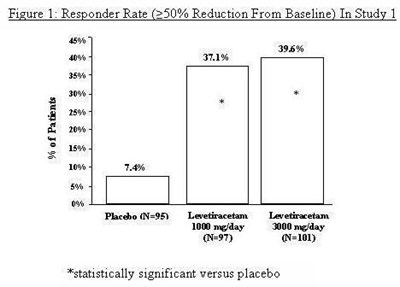
This is a graph that shows the responder rate (a 250 percent reduction from baseline) in Study 1, with percentages of patients on the Y-axis and the different treatments on the X-axis. There are three bars labeled placebo, levetiracetam 1000 mg/day, and levetiracetam 2000 mg/day. The graph shows that the responder rate is higher with both doses of levetiracetam compared to the placebo. The data indicate that the difference in responder rates is statistically significant versus placebo.*
Figure 2 - levetiracetam fig2
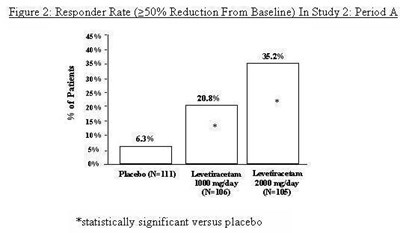
The figure shows the responder rate in Study 2 where there was a 250% reduction from baseline. The graph shows the percentage of patients who responded to treatment with PledioN-1l), Levetizacetam, or Levetiracetam at different dosage levels. The dosage levels for Levetiracetam were 100 mg/day and 2000 mg/day. The figure also mentions that the results were statistically significant compared to placebo.*
Figure 3 - levetiracetam fig3
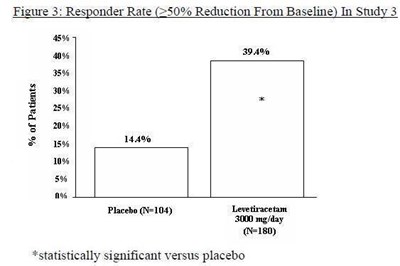
Figure 3 shows the responder rates, which measure the percentage of patients seeing a reduction of more than 50% from baseline, in Study 3. The graph displays the percentage of patients (from 0% to 144%) and the corresponding dosages of the drug Lewtiracstn (180 mg/day) and the placebo (N=104). The asterisk (*) indicates that the drug is statistically significant compared to the placebo.*
Figure 4 - levetiracetam fig4
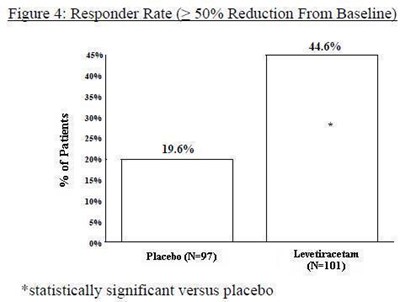
The figure 4 demonstrates the responder rate, which indicates the percentage of participants with more than 50% reduction in symptoms from baseline. The group labeled "B" had a responder rate of 44.6%, while the placebo group (N-97) had a responder rate of only 10.6%. The "B" group was statistically significant compared to the placebo group.*
Figure 5 - levetiracetam fig5

The figure shows the responder rate in terms of a 250% reduction from baseline in PGTC seizure frequency per week. The percentage of patients who achieved this reduction is represented on the Y-axis, ranging from 0% to 100%. The X-axis depicts the different groups of patients, with the placebo group (N=84) on the left and the levetiracetam group (N=79) on the right. The levetiracetam group achieved a statistically significant reduction in PGTC seizures compared to the placebo group.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
