Product Images Divalproex Sodium
View Photos of Packaging, Labels & Appearance
- 2ff00ecb deca 4c29 823b 6e76890b2869 01
- 2ff00ecb deca 4c29 823b 6e76890b2869 02
- 2ff00ecb deca 4c29 823b 6e76890b2869 03
- 2ff00ecb deca 4c29 823b 6e76890b2869 04
- 2ff00ecb deca 4c29 823b 6e76890b2869 05
- 2ff00ecb deca 4c29 823b 6e76890b2869 06
- 2ff00ecb deca 4c29 823b 6e76890b2869 07
- 2ff00ecb deca 4c29 823b 6e76890b2869 08
Product Label Images
The following 8 images provide visual information about the product associated with Divalproex Sodium NDC 68382-032 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
2ff00ecb deca 4c29 823b 6e76890b2869 03

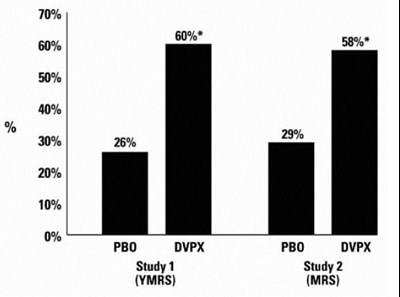
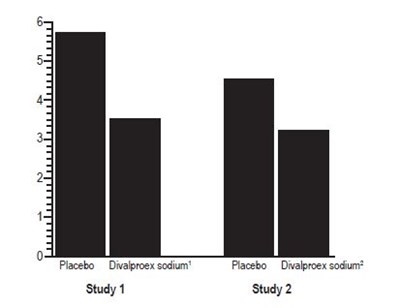
The text describes a graph or chart showing the reduction in CPS rate with the use of Divalproex Sodium Delayed-Release Tablets compared to a placebo. The chart also shows the percentage of patients who experienced improvement, no change, or worsening when using the drug.*
2ff00ecb deca 4c29 823b 6e76890b2869 06

This appears to be the label of a medication bottle for Divalproex Sodium delayed-release tablets. Unfortunately, the output seems to be incomplete and contains random characters, making it difficult to provide an accurate description.*
2ff00ecb deca 4c29 823b 6e76890b2869 07

This is a pharmaceutical label for Divalproex Sodium Delayed-release tablets, USP. The label includes dosage and prescription information.*
2ff00ecb deca 4c29 823b 6e76890b2869 08

This is a package of Divalproex Sodium tablets. It contains 100 delayed-release tablets with valproic acid activity. It is manufactured by Zydus Reony. The dosage and course of the medication is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.