Product Images Galantamine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 16 images provide visual information about the product associated with Galantamine NDC 68382-178 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
galantamine - 992cc7c5 4750 46be 8b7f d820000c8deb 01

Hydrogen bromide, commonly known as HBr, is a colorless gas used in the production of various chemicals including pharmaceuticals, flame retardants, and refrigerants. It is also used in the semiconductor industry as a cleaning agent. In aqueous solution, HBr forms hydrobromic acid.*
galantamine - 992cc7c5 4750 46be 8b7f d820000c8deb 03
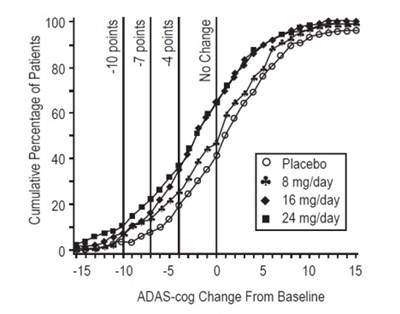
This appears to be a graph of the "Cumulative Percentage of Patients" with different doses of medication (8mg/day, 16mg/day, and 24mg/day) on the x-axis and the "ADAS-cog Change From Baseline" on the y-axis. The graph shows that there is no change in the ADAS-cog score for patients who took the placebo, while there is a decrease in ADAS-cog score for patients who took at least 8mg/day of medication. Not enough information is available to determine what the medication is or what ADAS-cog stands for.*
Galantamine - 992cc7c5 4750 46be 8b7f d820000c8deb 04

This appears to be a chart or table showing the percentage of patients and their respective treatments and outcomes. It is not clear what the specific conditions being treated are but there are four different treatments listed (placebo, 38mg/day, 16mg/day, 0.24mg/day) with corresponding columns indicating the percentage of patients marked as "Markedly Improved," "Moderately Improved," "Mainly - No Improved Change," "Minimally Worse," "Moderately Worse," and "Markedly Worse." The final column "CIBIC-plus Rating" is not clear what it refers to.*
galantamine - 992cc7c5 4750 46be 8b7f d820000c8deb 06

This is a graph showing the cumulative percentage of patients based on their ADAS-cog change from baseline. The graph displays the percentage of patients on the vertical axis and the change from baseline on the horizontal axis. The graph shows three different groups, including a placebo group, a 24mg/day group, and a 32mg/day group. The graph has a horizontal line at the 10 point mark indicating no change. Overall, the graph helps to visualize the effectiveness of the treatment being studied.*
galantamine - 992cc7c5 4750 46be 8b7f d820000c8deb 07

The text appears to be a graph showing the percentage of patients with different ratings and improvements for three different doses of a medication (placebo, 24mg/day, and 32mg/day) based on a CIBIC-plus rating. The graph also includes categories for "Minimaly Moderately Markedly" improved or worse. Therefore, it is a visualization of the efficacy of a medication at different doses.*
galantamine - 992cc7c5 4750 46be 8b7f d820000c8deb 08
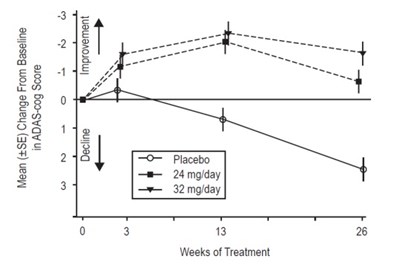
This appears to be a chart or table displaying the mean change from baseline in ADAS-cog score with different dosages of a treatment (24mg/day and 32mg/day) compared to a placebo. The chart illustrates the improvement seen after 13 weeks of treatment.*
galantamine Tablets USP, 8mg - 992cc7c5 4750 46be 8b7f d820000c8deb 15

The text appears to be a product description. It mentions a medication called Galantamine which comes in 60 tablets. The tablets are distributed by a company named Prmicesicds USA and it is a USP cvmreamamren. However, there is a typo in the text as some letters and characters are not recognizable.*
galantamine Tablets USP, 12mg - 992cc7c5 4750 46be 8b7f d820000c8deb 16

This is a description of a medication labeled as NDC 68382-17. Unfortunately, the text is incomplete and contains multiple errors and inconsistencies, including missing words, random characters, and incomplete sentences. Therefore, the description is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.







