Product Images Atorvastatin Calcium
View Photos of Packaging, Labels & Appearance
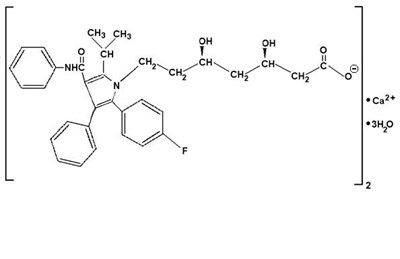
- Structured product formula for atorvastatin - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 01
- Structured product formula for atorvastatin - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 02
- Structured product formula for atorvastatin - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 03
- Structured product formula for atorvastatin - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 04
- Atorvastatin Calcium Tablets, 10 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 05
- Atorvastatin Calcium Tablets, 20 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 06
- Atorvastatin Calcium Tablets, 40 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 07
- Atorvastatin Calcium Tablets, 80 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 08
Product Label Images
The following 8 images provide visual information about the product associated with Atorvastatin Calcium NDC 68382-251 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Structured product formula for atorvastatin - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 02

The text consists of a chart displaying cumulative incidence percentages of a certain variable over a period of 35 years with two labels 'Atorvastatin' and 'Placebo'. There is also a number '84050089' and a couple of additional numbers which are not readable due to errors.*
Structured product formula for atorvastatin - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 03

The text represents a hazard analysis which shows the cumulative hazard percentage for certain primary endpoints over the course of four years. It also includes a hazard ratio of 0.63, with a confidence interval of 0.48-0.83 and a p-value of 0.001. However, there is no information given about the specific primary endpoints or any other details of the analysis.*
Structured product formula for atorvastatin - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 04

The text appears to be a table containing data on the percentage of subjects experiencing an event in two unspecified groups, as well as a result (HR 078) and its associated P value (0.0002). The table also includes a graph with four time points and a title indicating that it relates to major cardiovascular endpoints. No further context is available.*
Atorvastatin Calcium Tablets, 10 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 05

This is a description of a medication called Atorvastatin Calcium Tablets, with a strength of 10mg/tablet and a package size of 30 tablets. The tablets have a film coating and are distributed by Zydus Pharmaceuticals (USA) Inc. The usual dosage should be determined through consultation with a healthcare provider and the medication should be stored at a temperature between 20-25°C. The label provides detailed manufacturing and distribution information, and warns to keep the medication out of reach of children.*
Atorvastatin Calcium Tablets, 20 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 06

This is a label for a medication called Atorvastatin Calcium Tablets, which contains 20mg Atorvastatin Calcium per film-coated tablet. It has a usual dosage and should be stored at 20°C to 25°C. This medication should be kept out of reach of children. It is manufactured by Cadia Healthcare Ltd. from Ahmedabad, India, and distributed by Zydus Pharmaceuticals (USA) Inc. in Pennington, NJ.*
Atorvastatin Calcium Tablets, 40 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 07

This is a description of a drug package of Atorvastatin Calcium SR tablets manufactured by Cadia Healthcare Ltd. The package contains 30 tablets and has Lot number 000 and expiration date MMMYY. The tablets should be stored in controlled room temperature conditions and dispensed in a tight container. There is also a warning to keep the drugs out of reach of children. The package contains the NDC number 68382-251-06 and the tablets are for prescription use only.*
Atorvastatin Calcium Tablets, 80 mg - 27cfa7db 0057 40d0 9940 43f0dd0dbd57 08

This is a description for a medication called "Atorvastatin Calcium" which is presented in the form of tablets. The tablets contain 80mg of atorvastatin calcium, and are film-coated. The medication is available by prescription only and comes in a package of 30 tablets. The recommended storage is between 20°C to 25°C. The manufacturer of this medication is Cadia Healthcare Ltd in Ahmedabad, India and it is distributed by Zydus Pharmaceuticals (USA) Inc. in Pennington, NJ.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.