Product Images Bosentan
View Photos of Packaging, Labels & Appearance
- 041b3259 1370 4458 abef 2833886ade7e 01
- 041b3259 1370 4458 abef 2833886ade7e 02
- 041b3259 1370 4458 abef 2833886ade7e 03
- 041b3259 1370 4458 abef 2833886ade7e 04
- 041b3259 1370 4458 abef 2833886ade7e 05
- 041b3259 1370 4458 abef 2833886ade7e 06
- 041b3259 1370 4458 abef 2833886ade7e 07
- 041b3259 1370 4458 abef 2833886ade7e 08
- 041b3259 1370 4458 abef 2833886ade7e 09
- 041b3259 1370 4458 abef 2833886ade7e 10
Product Label Images
The following 10 images provide visual information about the product associated with Bosentan NDC 68382-447 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
041b3259 1370 4458 abef 2833886ade7e 01
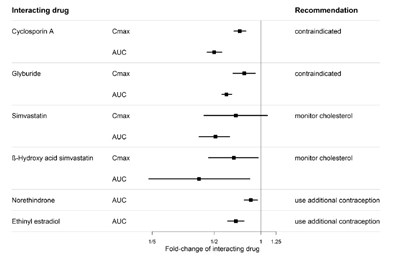
This is a description of several drugs and their interactions with each other. Cyclosporin A, Glyburide, Simvastatin, -Hydroxy acid simvastatin, Norethindrone, and Ethinyl estradiol are listed as interacting drugs. The text then shows values for Cmax and AUC. The fold-change of interacting drugs is also given, and a recommendation for each drug-interaction is listed. Cyclosporin A and Glyburide are contraindicated, Simvastatin and -Hydroxy acid simvastatin require cholesterol monitoring, and Norethindrone and Ethinyl estradiol require additional contraception.*
041b3259 1370 4458 abef 2833886ade7e 02

This text appears to be a chart showing different drugs and their interactions with enzymes, as well as measurements of their impact on the body's response to the drugs. The drugs mentioned are Cyclosporin A, Lopinavirritonavir, and Rifampin. The chart measures the "Ctrough Day 2," "Ctrough (ss)," "Cmax," and "AUC" of each drug at different time intervals. It also evaluates the "fold-change of bosentan." The chart recommends a dose reduction or weekly monitoring of AST/ALT for certain drugs and deems one drug contraindicated.*
041b3259 1370 4458 abef 2833886ade7e 04

This is a list of drugs and their interactions with other drugs that induce CYP3A or CYP2CY. It also includes recommended actions for healthcare providers based on the fold-change of the drug. Some drugs are contraindicated while others require monitoring of cholesterol levels or additional contraception. There are also Cmax and Auc values for some drugs available, but the context is not clear.*
041b3259 1370 4458 abef 2833886ade7e 05

This is a list of drugs and their effects on certain enzymes and transporters in the body. It also includes information on how these drugs affect the recommended dosage of another drug, bosentan. The text provides recommendations for dose adjustments, contraindications, and the fold-change of bosentan.*
041b3259 1370 4458 abef 2833886ade7e 07

The given text seems to be a graph that shows the effect of Bosentan on a particular parameter over a period of 28 weeks. However, without any axis labels or context, it is not possible to provide a useful description.*
041b3259 1370 4458 abef 2833886ade7e 08
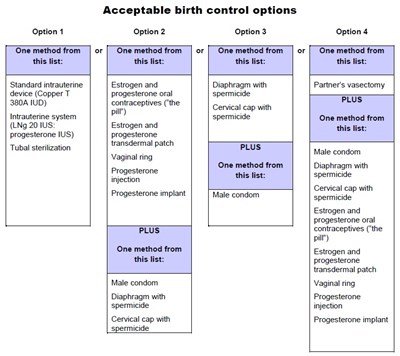
This is a list of acceptable birth control options which includes various methods such as intrauterine devices, hormonal contraceptives, diaphragms, spermicides, cervical caps, tubal sterilization, male condoms, progesterone implants and injections, and vaginal rings.*
041b3259 1370 4458 abef 2833886ade7e 09

This is a coding template for Bosentan tablets equivalent to 62.5 mg used for medication. The tablets should be stored at a temperature between 20°C to 25°C (68°F to 77°F) as per the USP controlled room temperature guidelines. The pack contains 100 tablets and is distributed by H Zydus Pharmaceuticals in the USA. The accompanying directions must be read carefully before use. It is essential to keep all medications out of the reach of children.*
041b3259 1370 4458 abef 2833886ade7e 10

This text seems to be a product label for Bosentan T tablets, an equivalent to 125mg of bosentan. The label includes information such as dosage, storage conditions, and precautions. The product seems to be manufactured in India by a company called Zydus.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

