Product Images Fesoterodine Fumarate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Fesoterodine Fumarate NDC 68382-479 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
cbef86b1 8a69 484d 87e8 05b6ecb06093 03

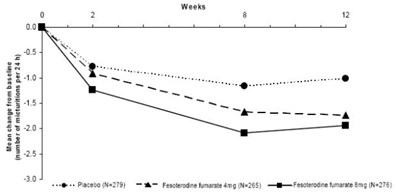
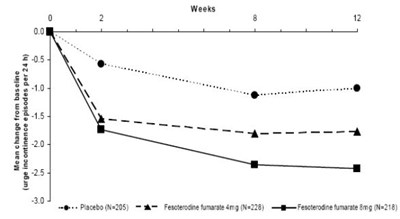
This is a chart displaying the change in urge incontinence episodes per 24 hours over the course of several weeks. The chart compares the effectiveness of a placebo (labeled as "Ne211"), a dosage of Fesoferodine fumarate (labeled as "Ne199"), and a higher dosage of Fesoferodine fumarate (labeled as "N223").*
cbef86b1 8a69 484d 87e8 05b6ecb06093 06

This is a drug package containing Fesoterodine Fumarate Extended-Release tablets of 4mg manufactured by Cadila Healthcare Ltd. The package contains 90 tablets and should be stored within the temperature range of 20°C to 25°C. The package has caution notes to protect from moisture and to keep it out of the reach of children. It is distributed by Zydus Pharmaceuticals in the USA and has a prescription-only label.*
cbef86b1 8a69 484d 87e8 05b6ecb06093 07

This is a medication with the NDC code 68382-480-16, containing 90 extended-release tablets of 8mg Fesoterodine Fumarate each, used for a specific indication not mentioned. It is distributed by Zydus Pharmaceuticals in the USA, and manufactured by Cadila Healthcare Ltd. in India. The package insert contains the recommended dosage and other prescription details. It should be stored at a controlled room temperature to protect from moisture and kept away from children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.