Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- structural formula image 01 - 4267f065 9c17 4df6 be68 f7d311f44a07 01
- Donepezil Tablet 23mg - 4267f065 9c17 4df6 be68 f7d311f44a07 02
- Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 03
- Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 04
- Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 05
- Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 06
- Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 07
- Donepezil Tablet 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 08
- Donepezil Tablet 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 09
- Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 10
- Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 11
- Figure 01 - 4267f065 9c17 4df6 be68 f7d311f44a07 12
- figure 02 - 4267f065 9c17 4df6 be68 f7d311f44a07 13
- figure 03 - 4267f065 9c17 4df6 be68 f7d311f44a07 14
- donepezilhacltabs-image05 - 4267f065 9c17 4df6 be68 f7d311f44a07 15
Product Label Images
The following 15 images provide visual information about the product associated with Donepezil Hydrochloride NDC 68382-521 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Donepezil Tablet 23mg - 4267f065 9c17 4df6 be68 f7d311f44a07 02

The text provides a chart indicating ADAS-cog (Alzheimer's Disease Assessment Scale-cognitive subscale) change from baseline after weeks of drug treatment with Donepezil Hydrochloride 10mg/day, Donepezil Hydrochloride 5mg/day, and placebo. It also indicates clinical improvement in percentage. No further information is available.*
Donepezil Tablets 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 05

This appears to be a graph showing the change in ADAS-cog score of patients after treatment with either Donepezil Hydrochloride (in 5mg or 10mg doses) or Placebo, over the course of several weeks. The graph shows that treatment with Donepezil Hydrochloride resulted in clinical improvement, while treatment with Placebo showed clinical decline. However, the text is not very clear and may contain errors.*
Donepezil Tablet 23 mg - 4267f065 9c17 4df6 be68 f7d311f44a07 09

The text appears to be a table with medication information, placebo, and baseline data. The medication listed is "Donepezil Hydrochloride" with a dosage of 10mg per day. The table also includes a placebo group with 30 participants and a baseline data column. It's unclear what the letters "E)" and "SIB" signify.*
Figure 01 - 4267f065 9c17 4df6 be68 f7d311f44a07 12
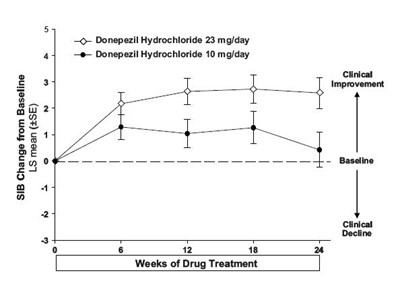
This appears to be a graph or chart showing the change from baseline in LS mean (SE) for Donepezil Hydrochloride at 23mg/day and 10mg/day over the course of 18 weeks of drug treatment. However, without additional context or information, it is difficult to provide a more specific or useful description.*
donepezilhacltabs-image05 - 4267f065 9c17 4df6 be68 f7d311f44a07 15

This is a medication package of Donepezil Hydrochloride Tablets with a concentration of 23 mg used in treating Alzheimer's disease. The medication is meant for oral administration only and comes with a separate patient package insert. The usual dosage is to be followed as per the prescribing information mentioned in the package insert. This medication should be stored at a temperature between 20° to 25°C and should be dispensed in a tight container. It is manufactured by Cadila Healthcare Ltd., Ahmedabad, India, and distributed by Zydus Pharmaceuticals USA Inc., Pennington, NJ 08534. The package contains 100 tablets, and it is strictly advised to keep this medication and all drugs out of the reach of children. The information is thus complete and reliable for users.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.









