Product Images Esomeprazole Magnesium
View Photos of Packaging, Labels & Appearance
- Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 01
- Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 02
- Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 03
- image - 2b944206 1301 4554 ba9a cf18f294f507 04
- image - 2b944206 1301 4554 ba9a cf18f294f507 05
- Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 06
- Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 07
- Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 08
- Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 09
Product Label Images
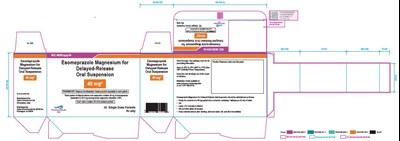
The following 9 images provide visual information about the product associated with Esomeprazole Magnesium NDC 68382-849 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 01

Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 02

Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 03

Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 06

Esomeprazole Magnesium For Delayed-Release Oral Suspension - 2b944206 1301 4554 ba9a cf18f294f507 07