Product Images Erlotinib
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Erlotinib NDC 68382-914 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - e715a711 283a 4662 8df0 d3b86ac5a042 02
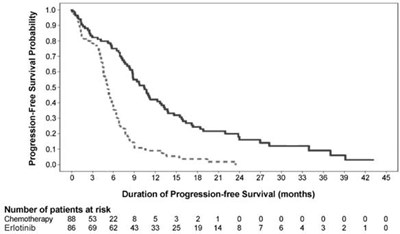
This appears to be a clinical study data table showing the Progression-Free Survival Probability of patients undergoing chemotherapy and Erlotinib treatment. It also provides the number of patients at risk in each case. The duration of Progression-free Survival is indicated in months.*
Image - e715a711 283a 4662 8df0 d3b86ac5a042 04

This text appears to be a chart or graph with data related to overall survival (in months) for two different groups of patients (Placebo and Erotinib) and the number of patients at risk in each group at different time points (o, 3, 6, 9, 12, 15, 18, 21, 24, and 27 months). Unfortunately, the text is not entirely clear due to errors generated by .*
Image - e715a711 283a 4662 8df0 d3b86ac5a042 05
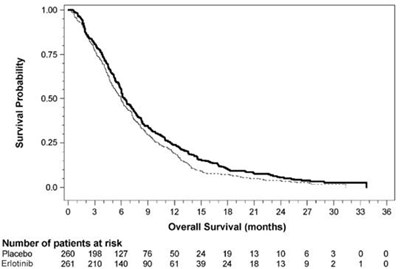
This appears to be a chart of the Survival Probability of patients taking either a placebo or Erlotinib, over a period of months. The chart shows the number of patients at risk for each group at different intervals. The survival probability is graphed along the y-axis from 0% to 100% and the overall survival time in months is along the x-axis.*
image - e715a711 283a 4662 8df0 d3b86ac5a042 06
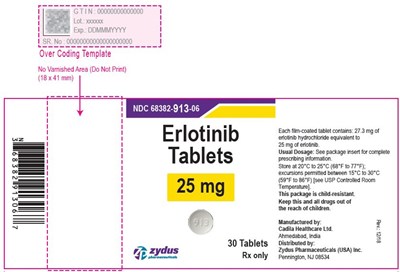
This appears to be a description of medication, specifically erlotinib tablets in 25mg dosage with a GTIN code of O and lot number X000. The tablets come in a package of 30 and are manufactured by Cadila Healthcare Ltd in India and distributed by Zydus Pharmaceuticals (USA) Inc in Pennington, NJ. The usual dosage information is not included in the text and is referred to the package insert or complete prescribing information. The tablets are film coated and the text includes information about storage temperature, controlled room temperature, and child-resistant packaging.*
image - e715a711 283a 4662 8df0 d3b86ac5a042 07
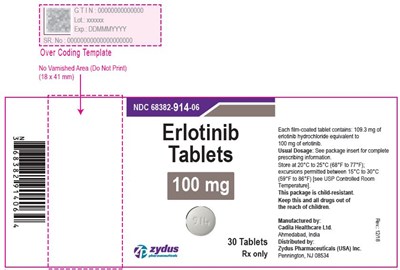
This is a description of a medication. It contains Erlotinib and comes in the form of film-coated tablets. Each tablet contains 100mg of Erlotinib G. The recommended storage temperature is between 20°C to 25°C or 68°F to T7°F with excursions permitted between 15°C 10 30°C or 59°F to 86°F. The package is child-resistant, and drugs should be kept out of the reach of children. The medication is manufactured by Cadia Healthcare Ltd in Abmedabad, India and distributed by Zydus Pharmaceuticals (USA) Inc.*
image - e715a711 283a 4662 8df0 d3b86ac5a042 08
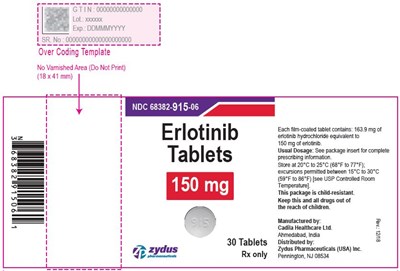
This is a description of a medication tablet package, specifically for Erlotinib tablets produced by Cadila Healthcare Ltd in India and distributed by Zydus Pharmaceuticals in the USA. The tablets come in a quantity of 30 and weigh 450mg each. The packaging includes a child-resistant mechanism, and the tablets should be stored within the temperature range of 20-25°C. Detailed prescribing information is provided in an enclosed package insert.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

