Product Images Natrum Muriaticum Kit Refill
View Photos of Packaging, Labels & Appearance
- image of vial/kit - Kit50 30c
- e6ffa829 610a 3393 e053 2995a90a9ec0
- e6ffb51c 402d 7c69 e053 2995a90aaa72
- e711ff0a 2fa2 1f2a e053 2a95a90a4e38
- e71249de 838b cb32 e053 2a95a90abc72
- e712591a 3530 7a17 e053 2995a90ad74b
- e712591a 3533 7a17 e053 2995a90ad74b
- e7128047 17ff 3426 e053 2995a90a3797
- e7128433 7d9b 5a16 e053 2a95a90ac7ed
- e712e417 4b7e 238e e053 2a95a90a292a
- e712ed8d bee5 4e43 e053 2995a90a8ef6
- e712ed8d bee7 4e43 e053 2995a90a8ef6
- e712fda9 504a 6d06 e053 2a95a90a61e4
- e7130907 6e84 74d2 e053 2995a90a082b
- e713547f b39b 237a e053 2995a90ae5eb
- e713695a df6c 0bc6 e053 2a95a90a31ba
- e7136efa b835 318d e053 2a95a90a961c
- e7137450 cae1 4055 e053 2995a90aaac3
- e71374f8 2ef3 42cf e053 2a95a90a105b
- e71374f8 2ef4 42cf e053 2a95a90a105b
- e7137f47 17ff 65ae e053 2a95a90a5a28
- e7139cc4 3b49 98e7 e053 2995a90ab860
- e713a100 1a9f 4399 e053 2a95a90aef61
- e713b537 757f 699a e053 2995a90a673a
- e713be46 70b3 10f3 e053 2995a90a7012
- e713d878 6caf ba56 e053 2995a90ab802
- e713f438 12e1 3eab e053 2995a90ac76d
- e714005f 8ccb 98b4 e053 2a95a90ad023
- e7140f60 e7e8 a072 e053 2a95a90a4308
- e7140f60 e7f8 a072 e053 2a95a90a4308
- e7144260 0e8d fded e053 2995a90a8114
- e714abd6 8468 c43a e053 2995a90a6956
- e714d613 e189 3242 e053 2995a90ab553
- e714f0f7 0942 74fc e053 2a95a90a0040
- e714fabf 20e7 2336 e053 2995a90a8edd
- e715081e 0a0b 3cd7 e053 2995a90aff94
- e715081e 0a0c 3cd7 e053 2995a90aff94
- e715081e 0a0e 3cd7 e053 2995a90aff94
- e7154011 d4f9 a827 e053 2995a90a64ec
- e715479c 73e7 3374 e053 2995a90a3ffa
- e715971f 2f2d 60e0 e053 2a95a90ae79d
- e7159e20 9ca6 06d9 e053 2995a90a5a0f
- e715b5f8 2635 6a90 e053 2995a90a3887
- e715ca41 92d1 da23 e053 2995a90abd95
- e715e037 dce9 3a32 e053 2995a90a21bf
- e715e037 dcea 3a32 e053 2995a90a21bf
- e715e30a 7151 44fb e053 2995a90a76d3
- e716057b dd5d 76f9 e053 2995a90addc0
Product Label Images
The following 48 images provide visual information about the product associated with Natrum Muriaticum Kit Refill NDC 68428-061 by Washington Homeopathic Products, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
e6ffa829 610a 3393 e053 2995a90a9ec0

This is a warning and directions label for a homeopathic medicine product with the active ingredient Aesculus Hipp 200c. The product contains sucrose/lactose pellets and is to be taken 3 times a day or as directed by a licensed practitioner. It is suggested to consult a practitioner if symptoms worsen or persist for more than 7 days or if pregnant or nursing. The product is not FDA evaluated.*
e6ffb51c 402d 7c69 e053 2995a90aaa72

This is a description of a homeopathic medicine by Washington Homeopathic products. The medicine contains sucrose/lactose pellets in which an extract of Aesculus Hipp is added, which is reportedly effective against hemorrhoids. The recommended dosage for adults or children is dissolving 3 to 5 pellets under the tongue three times a day as long as the symptoms persist but at greater intervals when it subsides. There is a warning if the symptoms worsen or persist for more than seven days, pregnant, or nursing especially while taking the medicine, consulting with a licensed practitioner is recommended. The ‘HPUS' letters indicate the component used in the product is officially monographed in the Homeopathic Pharmacopoeia of the United States. The product is not FDA evaluated, and the claims are based on traditional homeopathic practice.*
e711ff0a 2fa2 1f2a e053 2a95a90a4e38

This is a description of a homeopathic medicine produced by Washington Homeopathic Products, known for making homeopathic remedies since 1873. The medicine contains sucrose and lactose pellets and is called "WHE Allium Cepa 200¢". It claims to treat head colds based on traditional homeopathic practices, but has not been evaluated by the FDA. The letters “HPUS” indicate that the components of the product are officially monographed in the Homeopathic Pharmacopoeia of the United States. The article warns that users should not use the product if the seal is broken.*
e71249de 838b cb32 e053 2a95a90abc72

This text is a product label for Washington Homeopathic Products, Inc. The product is a homeopathic medicine that comes in pellet form containing sucrose/lactose. The label provides directions for adults or children to dissolve 3 to 5 pellets under the tongue, 3 times a day, or as directed by a licensed practitioner. It also includes a warning to consult a practitioner if symptoms worsen or persist after 7 days, or if the person is pregnant or nursing. The letters "HPUS" indicate that the product's component is officially monographed in the Homeopathic Pharmacopoeia of the United States. Additionally, the text includes the company's name, address, and website.*
e712591a 3530 7a17 e053 2995a90ad74b

This is a description of a homeopathic medicine in pellet form. The product contains Apis Mellifica 200C, a component that is officially monographed in the Homeopathic Pharmacopoeia of the United States (HPUS). The recommended dosage for adults and children is 3 times a day with 2 to 5 pellets dissolved under the tongue. The instructions advise consulting a licensed practitioner if symptoms persist for more than 7 days or if pregnant or nursing. The product is not evaluated by the FDA, and any claims are based on traditional homeopathic practice. The company selling the product is Washington Homeopathic Products, Inc., established since 1873.*
e712591a 3533 7a17 e053 2995a90ad74b

This is the label of a homeopathic product called "Apis Mellifica 30c" produced by Washington Homeopathic Products. The label warns that if symptoms worsen or persist for more than 7 days or if the person is pregnant or nursing, it is necessary to consult a practitioner. The product is not FDA evaluated, and claims are based on traditional homeopathic practice. The component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States; it uses sucrose/lactose pellets.*
e7128047 17ff 3426 e053 2995a90a3797

This is a warning and direction label of a homeopathic medicine product called Arnica Montana 30c by Washington Homeopathic Products. The product contains HPUS component and comes in the form of sucrose/lactose pellets. The label warns users to consult a licensed practitioner if symptoms worsen, persist for more than seven days, or if pregnant or nursing, and to keep out of reach of children. It also notes that claims are based on traditional homeopathic practice and not accepted medical evidence, and that the product should not be used if the seal is broken.*
e7128433 7d9b 5a16 e053 2a95a90ac7ed

This is a product description for a homeopathic medicine containing Arnica Montana 200c, used for the treatment of bruises. The medicine comes in sucrose/lactose pellets and is recommended to be taken by dissolving 3-5 pellets under the tongue, 3 times a day. The HPUS marking indicates that the component of the product is officially monographed in the Homeopathic Pharmacopoeia of the United States. The product claims are based on traditional homeopathic practice and have not been evaluated by the FDA. It is advised not to use the product if the seal is broken, and to keep it out of reach of children. If symptoms worsen or persist for more than 7 days or if pregnant or nursing, a licensed practitioner should be consulted.*
e712e417 4b7e 238e e053 2a95a90a292a
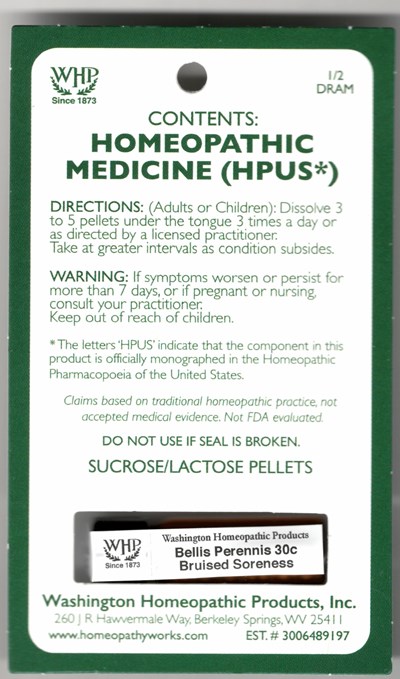
This is a homeopathic product warning label for Bellis Perennis 30c pellets for treating bruises and soreness. The label provides warning information such as consulting a practitioner if symptoms persist and keeping the product out of reach of children. The product contains sucrose and lactose pellets and is officially monographed in the Homeopathic Pharmacopoeia of the United States. The claims for this product are based on traditional homeopathic practice, not accepted medical evidence. This label is from Washington Homeopathic Products and is EST. # 3006489197.*
e712ed8d bee5 4e43 e053 2995a90a8ef6

This is a product warning for a homeopathic medicine manufactured by Washington Homeopathic Products, Inc. The warning advises adults and children to follow the provided instructions and to not use the product if the seal is broken. The medicine contains Sucrose/Lactose Pellets and Bellis Perennis 200c, which is used as a remedy for Bruised Soreness. The product is registered under the Homeopathic Pharmacopoeia of the United States (HPUS) and the website of the manufacturer is www.homeopathyworks.com.*
e712ed8d bee7 4e43 e053 2995a90a8ef6

This is a description for a homeopathic medicine product by Washington Homeopathic Products, Inc. The product contains Sucrose/Lactose Pellets and Onia Alba 200c that dissolves under the tongue. Users are advised to take 3 pellets 3 times a day as directed by a licensed practitioner, and to take the product at greater intervals as condition subsides. The letters "HPUS" indicates that the component in the product is officially monographed in the Homeopathic Pharmacopoeia of the United States. It's important to note that claims are based on traditional homeopathic practice and not FDA evaluated. The product should not be used if the seal is broken.*
e712fda9 504a 6d06 e053 2a95a90a61e4

This is a set of directions and warnings for using a homeopathic medication called Bryonia Alba 30c produced by Washington Homeopathic Products. The medication comes in the form of sucrose/lactose pellets, and users are instructed to dissolve 3-5 pellets under the tongue three times a day or as directed by a licensed practitioner. The warning provides advice to consult a practitioner if symptoms worsen or persist beyond seven days and advises pregnant and nursing individuals to seek medical advice. The text also contains information about the components of the homeopathic medicine and that the product is not FDA evaluated.*
e7130907 6e84 74d2 e053 2995a90a082b

This is a description of a homeopathic medicine produced by Washington Homeopathic Products. The medicine contains chamomilla 200¢c and is aimed at relieving irritability. The medicine should be dissolved under the tongue and taken three times a day or as directed by a licensed practitioner. There is a warning that if symptoms worsen or persist for more than 7 days, or if pregnant or nursing, one should consult their practitioner. The letters 'HPUS' indicate that the component in this product is officially monographed in the Homeopathic Medicine book. Claims for the medicine's effectiveness are based on traditional homeopathic practice, and it has not been evaluated by the FDA.*
e713547f b39b 237a e053 2995a90ae5eb

This is the product description and directions for a homeopathic medicine produced by Washington Homeopathic Products. The medicine comes in the form of sucrose/lactose pellets and the recommended dose is 3 pellets dissolved under the tongue, 3 times a day. It is important to consult a licensed practitioner before taking this medicine, especially if pregnant or nursing. The letters 'HPUS' indicate that the components in this product are officially listed in the Homeopathic Pharmacopoeia of the United States. Claims are based on traditional homeopathic practice and not on accepted medical evidence. If symptoms worsen or persist for more than 7 days, consult a practitioner.*
e713695a df6c 0bc6 e053 2a95a90a31ba

This is a product of homeopathic medicine, containing pellets that should be taken orally, 3 to 5 pellets, 3 times a day, under the tongue or as directed by a licensed practitioner. The pellets contain sucrose/lactose and the ingredient Hamamelis 200c, which is officially monographed in the Homeopathic Pharmacopoeia of the United States. The product claims are based on traditional homeopathic practice, not evaluated by the FDA. There is a warning to consult a practitioner if the symptoms worsen or persist for more than 7 days or if pregnant or nursing. Additionally, the product should not be used if the seal is broken and should be kept out of reach of children. The company Washington Homeopathic Products, Inc. has been in business since 1873 and is located in Berkeley Springs, WV. Their website is www.homeopathyworks.com.*
e7136efa b835 318d e053 2a95a90a961c

This is a description of a homeopathic medicine product from Washington Homeopathic Products, Inc. The product contains 30c of Cimicifuga Racemosa and is recommended for minor back pains. The medicine comes in sucrose/lactose pellets and the directions advise not to use if the seal is broken. The product is made following the guidelines of the Homeopathic Pharmacopoeia of the United States (HPUS).*
e7137450 cae1 4055 e053 2995a90aaac3

This is a description of a homeopathic medicine. The medicine comes in the form of pellets made of sucrose and lactose with the name Cimicifuga 200c, to relieve minor back pains. The product is officially monographed in the Homeopathic Pharmacopoeia of the United States as indicated by the letters 'HPUS'. The medicine should be dissolved under the tongue three times a day by adults or children, as directed by a licensed practitioner. The warning advises that if symptoms worsen or persist for more than 7 days, or if pregnant or nursing, consult your practitioner. The product is not evaluated by FDA, and the claims are based on traditional homeopathic practice. Keep the medicine out of reach of children, and do not use it if the seal is broken. The manufacturer's address is Woashington Homeopathic Products, Inc., 260 | R Hawvermale Way, Berkeley Springs, WV 2541 | www.homeopathyworks.com EST.# 3006489197.*
e71374f8 2ef3 42cf e053 2a95a90a105b

This is a homeopathic medicine (HPUS) named Eupatorium Pert which is designed to help alleviate flu-like symptoms. Adults and children are instructed to dissolve 3-5 pellets under the tongue three times a day, with greater intervals when feeling better. It is advised to consult a licensed practitioner if symptoms worsen or persist for more than 7 days or if pregnant or nursing. The product contains sucrose/lactose pellets and the letters "HPUS" indicate that the component is in the Pharmacopoeia of the United States. The effectiveness of the product is based on traditional homeopathic practice and not FDA evaluated. It is advised to not use the product if the seal is broken.*
e71374f8 2ef4 42cf e053 2a95a90a105b

This is a warning label for a homeopathic medicine called "Eupatorium Perf 200c" manufactured by Washington Homeopathic Products. The medicine consists of pellets made of sucrose and lactose that are dissolved under the tongue 3 times a day, as per the instruction given in the label. The medicine is indicated to treat flu-like symptoms. The label also warns against the worsening of symptoms or the persistence of such symptoms for more than 7 days, and further advises those who are pregnant or nursing to seek consultation with a licensed practitioner before using the product. The text also mentions that the component in the product is officially monographed in the Homeopathic Pharmacopoeia of the United States, but the claims based on traditional homeopathic practice are not evaluated by FDA.*
e7137f47 17ff 65ae e053 2a95a90a5a28

This is a homeopathic medicine labeled as "Coffea Cruda 200c" which is primarily used to relieve symptoms of sleeplessness. The product should be dissolved to five pellets under the tongue three times a day or as directed by a licensed practitioner. The warning suggests consulting a practitioner before using during pregnancy or nursing. In case of worsened symptoms or persisting symptoms after 7 days, a practitioner should be consulted. The product is made of sucrose/lactose pellets and should be kept out of children's reach. The product claims are based only on traditional homeopathic practices and is not FDA evaluated. The monographed component of this product is officially approved by HPUS. The manufacturer of the product is Washington Homeopathic Products established in 1873.*
e7139cc4 3b49 98e7 e053 2995a90ab860

This is a homeopathic medicine that contains pellets made of sucrose and lactose. The medicine is named "Euphrasia Officinalis 30c" and can be dissolved under the tongue. The recommended dose for adults and children is 3 times a day, or as instructed by a licensed practitioner. The warning mentions that if the symptoms worsen or persist for more than 7 days or one is pregnant, they should consult the practitioner. The product is not FDA evaluated and does not have any claims based on medical evidence.*
e713a100 1a9f 4399 e053 2a95a90aef61

This is a warning and instruction label for a Homeopathic medicine sold by Washington Homeopathic Products. The medicine is designed to treat sleeplessness and comes in small Sucrose/Lactose pellets. Consumers are directed to dissolve 3 to 5 pellets under their tongue three times a day or as directed by a licensed practitioner. It is important to note that if symptoms worsen or persist for more than 7 days, or if pregnant or nursing, consumers must consult their healthcare practitioner. The letters ‘HPUS' indicate that the medicine's component is officially monographed in the Homeopathic Pharmacopoeia of the United States. The claims are based on traditional homeopathic practice and not accepted medical evidence. The product is not FDA evaluated. The product should not be used if the seal is broken, and it should be kept out of reach of children.*
e713b537 757f 699a e053 2995a90a673a

This is a package of homeopathic medicine from Washington Homeopathic Products designed to help with cough. The medicine contains pellets made out of sugar (sucrose) and lactose that need to be dissolved under the tongue by adults or children up to 5 times per day or as directed by a physician. The letters 'HPUS' indicate the product's components are officially monographed in the Homeopathic Pharmacopoeia of the United States. The claims regarding the product's efficacy are not based on accepted medical evidence but rather on traditional homeopathic practice. It is important to keep the product out of reach of children, and if the symptoms worsen or persist for more than 7 days, or if the consumer is pregnant or nursing, it is recommended to seek medical advice from a practitioner.*
e713be46 70b3 10f3 e053 2995a90a7012

This is a description of a homeopathic medicine by Washington Homeopathic Products. The medicine contains sucrose/lactose pellets and is indicated for the treatment of cough. The user is instructed to dissolve 3 to 5 pellets under the tongue three times a day or as directed by a licensed practitioner. It is important to seek medical advice if symptoms worsen or persist for more than 7 days, or if the user is pregnant or nursing. The medicine is not evaluated by FDA, and claims are based on traditional homeopathic practice. The product must not be used if the seal is broken. The letters "HPUS" indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States.*
e713d878 6caf ba56 e053 2995a90ab802

This is a product label for a homeopathic remedy for eye irritation containing sucrose/lactose pellets with an ingredient called "Euphrasia Officinalis". It includes a warning to consult with a practitioner if symptoms persist or if pregnant/nursing, a precaution to keep out of reach of children, and a disclaimer that the claims have not been evaluated by FDA. The product is produced by a company called Washington Homeopathic Products, Inc.*
e713f438 12e1 3eab e053 2995a90ac76d

This is a direction for using a homeopathic medicine called Hamamelis Virginiana 30c, which is indicated for treating varicose veins. The medicine comes in the form of pellets, and the recommended dosage for both adults and children is between 3 to 5 pellets taken orally thrice daily or as directed by a licensed health practitioner. The medicine should be taken at greater intervals as the condition improves. It is also advised to consult a doctor if the symptoms persist after seven days or if you are pregnant or nursing. The medicine should be kept out of reach of children, and one should not use it if the seal is broken. The text contains information about harmless traditional homeopathic practices and mentions that the letters "HPUS" indicate the component in the Pharmacopoeia of the United States. The text warns that the product does not have FDA evaluation.*
e714005f 8ccb 98b4 e053 2a95a90ad023

This is a description of a homeopathic medicine, with instructions for use and warning for if symptoms worsen or persist. It is a sucrose/lactose pellet containing Hypericum 30c, used for shooting pain relief. The medicine is officially monographed in the Homeopathic Pharmacopoeia of the United States, with claims based on traditional homeopathic practice and not yet evaluated by the FDA. This product is manufactured and distributed by Washington Homeopathic Products, Inc.*
e7140f60 e7e8 a072 e053 2a95a90a4308
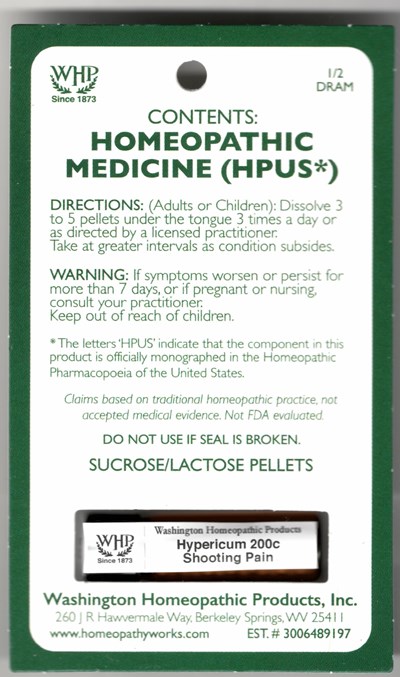
This is a description of a homeopathic medicine named "Hypericum 200c" produced by Washington Homeopathic Products. The recommended dosage for adults and children is 3 to 5 pellets under the tongue, 3 times a day, as advised by a licensed practitioner. The product is made according to the standards of Homeopathic Pharmacopoeia of the United States as indicated by the letters 'HPUS'. While claims are based on traditional homeopathic practices, the product has not been evaluated by the FDA and is not backed by accepted medical evidence. The medicine comes in a sucrose/lactose pellet form and should not be used if the seal is broken. Information on how to purchase the product is available on the company's website.*
e7140f60 e7f8 a072 e053 2a95a90a4308

This is a product label for a homeopathic medicine by Washington Homeopathic Products, Inc. The medicine is Kali Bichromicum 200c for sinus issues. The label provides instructions for use in adults or children, with 3 pellets to be taken under the tongue 3 times a day or as directed by a licensed practitioner. The text mentions that the letters 'HPUS' indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States. It also mentions that the claims for the product are based on traditional homeopathic practice and have not been evaluated by the FDA.*
e7144260 0e8d fded e053 2995a90a8114

This is a homeopathic product called Kali Bichromicum 30c intended for relieving sinus symptoms. The recommended dosage is 3 times a day with 1 to 5 pellets under the tongue. It is important to consult with a licensed practitioner before using this product, to take it at greater intervals if the condition worsens or if one is pregnant or nursing. The product is not FDA evaluated and should not be used if the seal is broken. The label includes the company information and a warning to keep out of reach of children.*
e714abd6 8468 c43a e053 2995a90a6956

This is a homeopathic medicine containing pellets made of sucrose/lactose. The medicine is to be taken by dissolving 3 to 5 pellets under the tongue three times per day or as directed by a licensed practitioner. It is advised to take at greater intervals as the condition subsides. The product is officially monographed in the Homeopathic Pharmacopoeia of the United States. The claims are based on traditional homeopathic practice and have not been evaluated by the FDA. The component of this product is Kali Carbonicum 200c which is used for sour belching since 1673. A warning is given to consult a practitioner if symptoms worsen or persist after 7 days, or if pregnant or nursing. The product should be kept out of reach of children, and not used if the seal is broken.*
e714d613 e189 3242 e053 2995a90ab553

This is a homeopathic medicine called WHY ® DRAM which comes in the form of sucrose/lactose pellets that should be dissolved under the tongue. The dosage is 3 to 5 pellets, 3 times a day, or as directed by a licensed practitioner. The letters "HPUS" indicate that the component of this product is officially monographed in the Homeopathic Pharmacopoeia of the United States. The product claims are based on traditional homeopathic practice, not accepted medical evidence, and it is not FDA evaluated. It should not be used if the seal is broken and should be kept out of reach of children. This product is made by Washington Homeopathic Products and contains Kali Carbonicum 30c. The product targets sour belching, and if symptoms worsen or persist for more than 7 days, or if pregnant or nursing, a practitioner should be consulted.*
e714f0f7 0942 74fc e053 2a95a90a0040

This is a description of a homeopathic medicine called "Natrum Muriaticum 30c" that comes in the form of pellets made of sucrose/lactose. The medicine is indicated for both adults and children, and the recommended dosage is 3 times a day with up to 5 pellets under the tongue. However, it is advised to consult with a licensed practitioner and to stop using it if symptoms worsen, persist for more than 7 days, or in case of pregnancy or nursing. This medicine is officially monographed in the Homeopathic Pharmacopoeia of the United States. The available information is based on traditional homeopathic practice, not on accepted medical evidence.*
e714fabf 20e7 2336 e053 2995a90a8edd

This text is a description of a homeopathic medicine called Natrum Muriaticum 200c, made by Washington Homeopathic Products. The medicine comes in the form of pellets made of sucrose/lactose and is meant to be dissolved under the tongue, 3 times a day, as directed by a licensed practitioner. The text warns users to consult their practitioner if symptoms worsen, persist for more than 7 days, or if pregnant or nursing. The medicine has not been evaluated by the FDA and claims are based only on traditional homeopathic practice, not accepted medical evidence.*
e715081e 0a0b 3cd7 e053 2995a90aff94

This is a description for a homeopathic medicine product that contains Sucrose/Lactose Pellets and Podophyllum Peit 30c. The medicine is to be taken as directed by a licensed practitioner and dissolves 3 to 5 pellets under the tongue 3 times a day. The warning section cautions against worsening of symptoms or persisting symptoms for more than 7 days, and using the product while pregnant/nursing. It mentions that the product is officially monographed in the Homeopathic Pharmacopoeia of the United States and not FDA evaluated. The product is manufactured by Washington Homeopathic Products since 1873 and its address and website are provided.*
e715081e 0a0c 3cd7 e053 2995a90aff94

This is a description of a homeopathic medicine product named "Podaphylium Peit 200¢" manufactured by Washington Homeopathic Products, Inc. The medicine is in pellet form that needs to be dissolved under the tongue three times a day or following the instructions of a licensed practitioner. The product is not FDA evaluated and should not be used if the seal is broken. The text also includes a warning to consult a practitioner in case of persistent symptoms or pregnancy/nursing. It is claimed to be based on traditional homeopathic practice and contains sucrose/lactose pellets. The inclusion of HPUS indicates that the component in the product is officially monographed in the Homeopathic Pharmacopoeia of the United States.*
e715081e 0a0e 3cd7 e053 2995a90aff94

This is a description of a homeopathic medicine product made by Washington Homeopathic Products. The medicine is in pellet form, and comes in a bottle with a broken seal warning. The product contains sucrose/lactose pellets, and the recommended dosage for adults or children is to dissolve 5 pellets under the tongue 3 times a day, as directed by a licensed practitioner. The product should be kept out of reach of children, and adults experiencing worsening symptoms after 7 days or while pregnant or nursing should consult a practitioner. The product claims are based on traditional homeopathic practice and are not evaluated by the FDA.*
e7154011 d4f9 a827 e053 2995a90a64ec

This is a description of a homeopathic medicine product marketed by Washington Homeopathic Products. The product is in the form of pellets and is intended for adults or children. The medication is dissolved under the tongue three times a day as directed by a licensed practitioner. The medicine is not FDA evaluated, and the letters 'HPUS' indicate that the component is officially monographed in the Homeopathic Pharmacopoeia of the United States. The product claims its efficacy based on traditional homeopathic practices. If symptoms worsen or persist for more than seven days, it is recommended to consult a practitioner.*
e715479c 73e7 3374 e053 2995a90a3ffa

This is a warning statement and description for a homeopathic medicine. It includes instructions for use, a warning about worsening symptoms, and caution for use during pregnancy or nursing. The product is only based on traditional homeopathic practice and not evaluated by the FDA. The product contains sucrose/lactose pellets and a component officially monographed in the Homeopathic Pharmacopoeia of the United States. The manufacturer is Washington Homeopathic Products, established in 1873.*
e715971f 2f2d 60e0 e053 2a95a90ae79d

This appears to be the description of a homeopathic medicine product by Washington Homeopathic Products, founded in 1873. The product, WHE Sulphur 200c, is made with sucrose/lactose pellets and is indicated for treating skin problems. The letters "HPUS" indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States. However, claims based on traditional homeopathic practice and there is no FDA evaluation.*
e7159e20 9ca6 06d9 e053 2995a90a5a0f

This is a description for a homeopathic medicine product from Washington Homeopathic Products. The product contains Ruta Graveolens 200c and has been used for the relief of bruised feelings since 1873. The medicine is made up of sucrose/lactose pellets and is officially monographed in the Homeopathic Pharmacopeia of the United States (HPUS). Users must consult with a practitioner if symptoms worsen or persist for more than 7 days, or if pregnant or nursing. The product is not evaluated by the FDA, and its claims are based on traditional homeopathic practice. It is also emphasized not to use the product if the seal is broken and to keep it out of reach of children.*
e715b5f8 2635 6a90 e053 2995a90a3887

This is a homeopathic medicine product and the directions for use indicate that adults or children should dissolve 3 to 5 pellets under the tongue three times per day. It also includes a warning indicating that if symptoms worsen or persist, or if pregnant or nursing, to consult a practitioner, and to keep the product out of the reach of children. The product is officially monographed in the Homeopathic Pharmacopoeia of the United States. The claims are based on traditional homeopathic practice and the product has not been FDA evaluated. The product contains Sucrose/Lactose pellets and the active ingredient is Staphysagria 30c. The manufacturer is Washington Homeopathic Products, Inc., and the address and website are provided.*
e715ca41 92d1 da23 e053 2995a90abd95

This is a description of a homeopathic medicine produced by Washington Homeopathic Products company. The product, named "Sulphur 30c", is intended for treating skin problems. The label provides directions for use: dissolve 3 to 5 pellets under the tongue 3 times a day or as directed by a licensed practitioner. The warning states that if symptoms worsen or persist for more than 7 days, or if pregnant or nursing, a doctor should be consulted. It also reminds users that the product should be kept out of reach of children, and not to be consumed if the seal is broken. The text emphasizes that the product's component is officially monographed in the Homeopathic Pharmacopoeia of the United States. Additionally, the claims about the medicine are based on traditional homeopathic practice, not accepted medical evidence, and are not evaluated by the FDA. Note that there is a typo in the flyer, the word "since" should be replaced by "since".*
e715e037 dce9 3a32 e053 2995a90a21bf

This is the product information for a homeopathic medicine with Symphytum Off 200 as its active ingredient. It includes directions for use, warnings, and information on the product's compliance with the Homeopathic Pharmacopoeia of the United States. Claims regarding the medicine are based on traditional homeopathic practice and have not been evaluated by the FDA.*
e715e037 dcea 3a32 e053 2995a90a21bf

This text appears to be a set of directions for a homeopathic medicine product called "Symphytum c" by Washington Homeopathic Products. The medicine is to be dissolved as pellets under the tongue three times a day, and if symptoms worsen or persist for more than 7 days, it is recommended to consult a licensed practitioner. The text also includes a warning to keep the product out of reach of children and notes that the medicine's components are officially monographed in the Homeopathic Pharmacopoeia of the United States. The claims of the effectiveness of the product are based on traditional homeopathic practices and are not evaluated by the FDA.*
e716057b dd5d 76f9 e053 2995a90addc0

This is a description of a homeopathic medicine product containing Sucrose/Lactose Pellets with Thuja Occidentalis 30c as an active ingredient. It provides directions for use, warnings, and cautions. The component of the product is officially listed in the Homeopathic Pharmacopoeia of the United States. The claims are based on traditional homeopathic practice and not FDA evaluated. The product is from Washington Homeopathic Products Inc. and has been in the market since 1873.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

