FDA Label for Pca Skin Skin Procedure
View Indications, Usage & Precautions
Pca Skin Skin Procedure Product Label
The following document was submitted to the FDA by the labeler of this product Cp Skin Health Group, Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
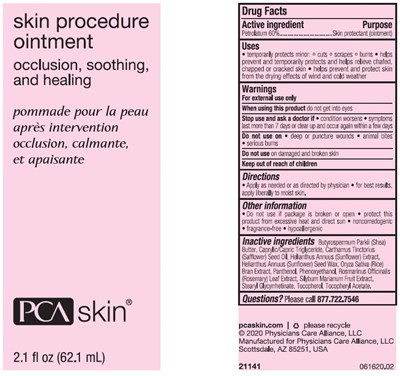
Active Ingredient
Petrolatum 60%
Purpose
Skin protectant (ointment)
Uses
• temporarily protects minor: ° cuts ° scrapes ° burns • helps prevent and temporarily protects and helps relieve chafed, chapped or cracked skin • helps prevent and protect skin from the drying effects of wind and cold weather
Warnings
For external use only
When Using This Product
do not get into eyes
Stop Use And Ask A Doctor If
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Do Not Use On
- deep or puncture wounds
- animal bites
- serious burns
Do not use on damaged and broken skin
Directions
- Apply as needed or as directed by physician
- for best results, apply liberally to moist skin.
Other Information
- Do not use if package is broken or open
- protect this product from excessive heat and direct sun
- noncomedogenic
- fragrance-free
- hypoallergenic
Inactive Ingredients
Butyrospermum Parkii (Shea) Butter, Caprylic/Capric Triglyceride, Carthamus Tincotirius (Safflower) Seed Oil, Helianthus Annuus (Sunflower) Extract, Helianthus Annuus (Sunflower) Seed Wax, Oryza Sativa (Rice) Bran Extract, Panthenol, Phenoxyethanol, Rosamarinus Officinalis (Rosemary) Leaf Extract, Silybum Marianum Fruit Extract, Stearyl Glycyrrhetinate, Tocopherol, Tocopheryl Acetate.
Questions?
Please call 877.722.7546
Package Labeling:
* Please review the disclaimer below.