Product Images Alendronate Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Alendronate Sodium NDC 68788-7130 by Preferred Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
2 - 1203dd0c 809a 4409 b948 b5e816b8a63a 02
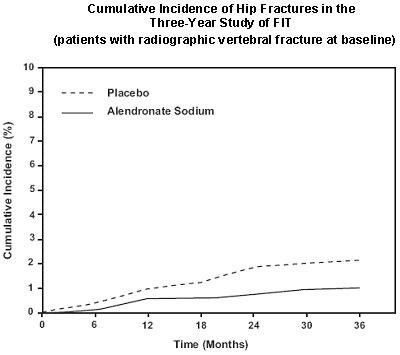
This is a graph displaying the cumulative incidence of hip fractures over a 3-year study of FIT, focused on patients with radiographic vertebral fracture at baseline. On the x-axis there is a timeline displayed in months, and on the y-axis there is a percentage scale ranging from 0% to 12%. There are two lines plotted: one represents the placebo group, and the other represents the alendronate sodium group.*
4 - 1203dd0c 809a 4409 b948 b5e816b8a63a 04

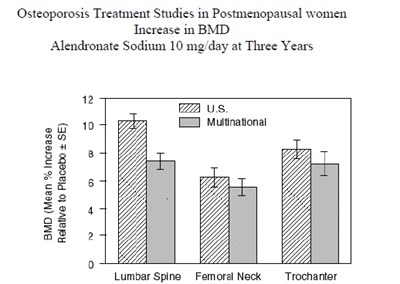
Osteoporosis Treatment Studies have been conducted in postmenopausal women, comparing the effect of Alendronate Sodium 10 mg/day versus a placebo. The studies track the time course of the change in Lumbar Spine BMD (bone mineral density) in terms of percentage change from baseline. The data is presented in two sets: a US Study and a Multinational Study. The study duration is represented in months. Unfortunately, the text contains some gibberish characters making it difficult to interpret.*
7 - 1203dd0c 809a 4409 b948 b5e816b8a63a 07
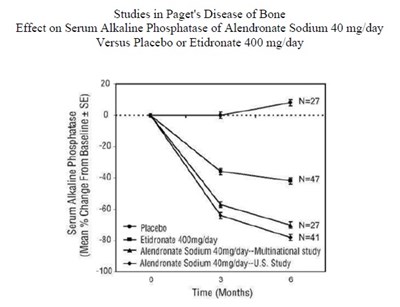
This is a study comparing the effect of Alendronate Sodium 40mg/day vs placebo or Etidronate 400mg/day in patients with Paget's Disease of Bone. The study has a sample size of 27 or 47 individuals and investigates serum alkaline phosphatase levels over time. The results of the study are presented graphically using mean percent change from baseline with standard error bars.*
Alendronate Sodium Tablets, USP 70mg - 1203dd0c 809a 4409 b948 b5e816b8a63a 09

This is a description of Alendronate Sodium tablets, which is the generic drug for Fosamax. Each tablet contains 91 37mg of alendronate sodium thyrate. The recommended dose of the drug should be taken as directed and caution should be taken as it is prohibited by federal law to transfer the drug to any other person than the one it was prescribed to. The manufacturer is Cipla USA. The text also contains product numbers, lot numbers and insurance NDCs.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.