Product Images Nifedipine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Nifedipine NDC 68788-7642 by Preferred Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
02 - image 02
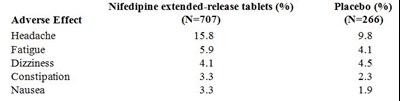
The given text mentions the adverse effects of Nifedipine extended-release tablets including headache, fatigue, dizziness, constipation, and nausea along with the percentage of occurrence among 707 patients. It also provides the corresponding percentage of patients experiencing similar effects in the placebo group consisting of 266 individuals.*
03 - image 03

This is a table comparing the percentage of adverse effects experienced by patients taking Nifedipine capsules versus a placebo. The adverse effects stated include dizziness, lightheadedness, giddiness, flushing, heat sensation, headache, weakness, nausea, heartburn, muscle cramps, tremors, peripheral edema, nervousness, mood changes, palpitations, dyspnea, cough, wheezing, nasal congestion, and sore throat. The sample size of patients is given for both Nifedipine and placebo groups.*
Nifedipine Extended Release Tablet USP 30mg - image 05

Nifedipine Extended Release Tab is a medication used for treating high blood pressure. The tablets contain 30mg of the active ingredient, nifedipine. The medication comes in a packet with an expiration date, batch and lot number. The manufacturer is Ingenus Pharmaceuticals, LLC.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

