Product Images Duloxetine
View Photos of Packaging, Labels & Appearance
- Duloxetine Delayed Release Cap USP 30mg
- figure1 - duloxetine fig1
- figure10 - duloxetine fig10
- figure2 - duloxetine fig2
- figure3 - duloxetine fig3
- figure4 - duloxetine fig4
- figure5 - duloxetine fig5
- figure6 - duloxetine fig6
- figure8 - duloxetine fig8
- figure9 - duloxetine fig9
- structure - duloxetine structure
Product Label Images
The following 11 images provide visual information about the product associated with Duloxetine NDC 68788-7935 by Preferred Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Duloxetine Delayed Release Cap USP 30mg

This is a description of a medication named Duloxetine Delayed-Release Cap. It is a USP 30mg capsule that contains Duloxetine. The manufacturer is Ajanta Pharma USA Inc, and the product is a generic alternative for Cymbalta. The document also contains information such as lot number, package size, expiration date, and cautions about the federal law.*
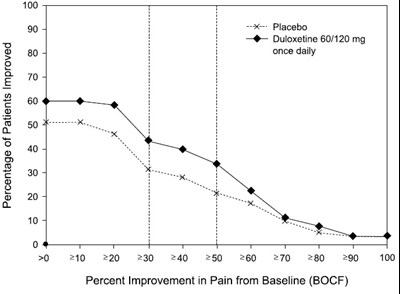
figure10 - duloxetine fig10

This is a description of a medication regimen for pain relief involving the drug Duloxetine, which is taken once a day in doses of 60 or 120mg. The document includes a chart indicating improvement in pain levels over time as a percentage.*
figure2 - duloxetine fig2
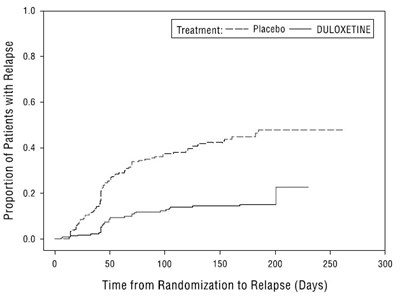
The text above is a graph showing the proportion of patients with relapse over time from randomization to relapse, for two treatments: Placebo and Duloxetine. The x-axis represents the time from randomization to relapse (in days), while the y-axis shows the proportion of patients with relapse (from 0 to 10%).*
figure3 - duloxetine fig3
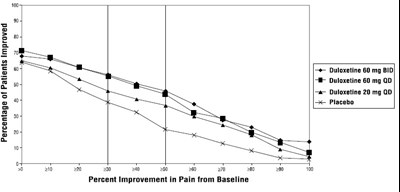
This is a table showing the percentage of patients improved for different doses of Duloxetine (60mg BID, 60mg QD, and 20mg QD) in comparison to placebo in terms of pain improvement from baseline.*
figure5 - duloxetine fig5

This is a table showing the percentage of patients who improved based on different treatments for pain. The treatments listed are Dulozetine 60mg twice daily, Duloxetine 60mg once daily, and Placebo. The table also shows the percent improvement in pain from baseline.*
figure8 - duloxetine fig8
This text describes a medication called Duloxetine, which is taken once daily at a dose of either 60mg or 120mg. The name "Placebo" is mentioned but it is unclear how it relates to the medication. There is a table or graph labeled "Percent Improvement in Pain from Baseline (BOCF)" but no additional information is provided about it. The final line of text appears to be gibberish and may have been affected by the .*
figure9 - duloxetine fig9
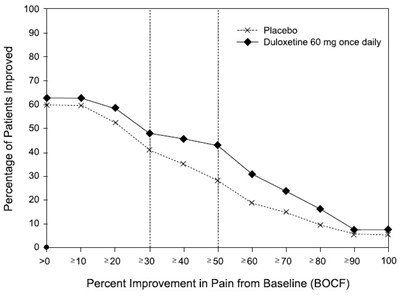
This is a report on the effectiveness of Placebo and Duloxetine 60 mg at reducing pain. The report presents a graph of percent improvement in pain from baseline, with different time intervals represented on the x-axis, and percent improvement on the y-axis. The text also includes some numbers and symbols that are not easily interpreted.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



