Product Images Zolpidem Tartrate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Zolpidem Tartrate NDC 68788-8118 by Preferred Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - zolpidem 02
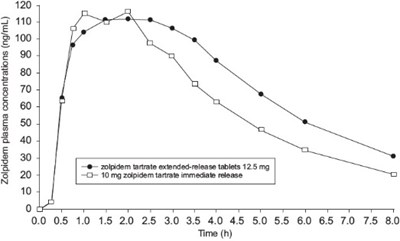
This is a graph that shows the plasma concentrations of zolpidem, a sleep medication, in ng/mL over a period of 8 hours after taking extended-release tablets of 12.5 g or immediate-release tablets of 10 mg. The x-axis represents time in hours and the y-axis represents the plasma concentration in ng/mL.*
Zolpidem Tartrate ER Tab 625mg CIV - zolpidem 03

This is a description of a medication called Zolpidem Tartrate ER Tab. It is a generic version of a medication called Ambien. The tablet is an extended-release tablet that contains 6.25mg of Zolpidem Tartrate ER. It is manufactured by Breckenridge Pharmaceuticals in Boca Raton, Florida. There are cautionary statements on the bottle that prohibit the transfer of this drug to anyone other than the patient for whom it was prescribed. The package size, expiration date, lot number, batch number, and prod number are all listed. There are several repeated entries for Quy, Insurance, Lot number, Batch number, and Prod number, but no actual information is given about their values for this particular medication. The text ends abruptly after a log chart.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.