Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
- Figure 1: Effect of interacting drugs on the pharmacokinetics of venlafaxine and active metabolite O-desmethylvenlafaxine (ODV). - venlafaxine fig1
- Figure 2: Effect of venlafaxine on the pharmacokinetics interacting drugs and their active metabolites. - venlafaxine fig2
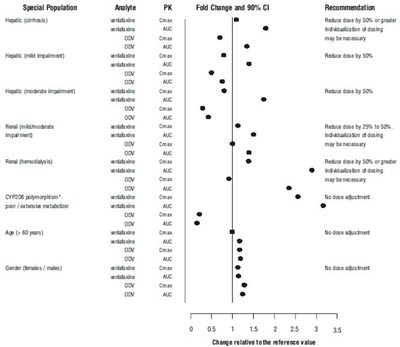
- Figure 3: Pharmacokinetics of venlafaxine and its metabolite O-desmethylvenlafaxine (ODV) in special populations. - venlafaxine fig3
- Venlafaxine HCl Extended-Release Capsules 75mg - venlafaxine fig4
- Chemical Structure - venlafaxine str
Product Label Images
The following 5 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 68788-8140 by Preferred Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Effect of interacting drugs on the pharmacokinetics of venlafaxine and active metabolite O-desmethylvenlafaxine (ODV). - venlafaxine fig1
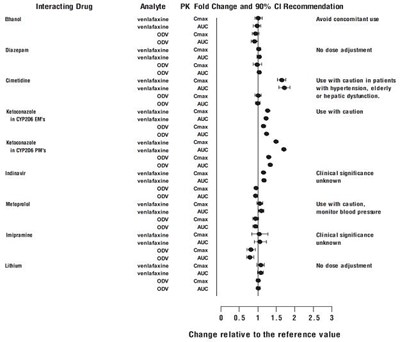
Figure 2: Effect of venlafaxine on the pharmacokinetics interacting drugs and their active metabolites. - venlafaxine fig2
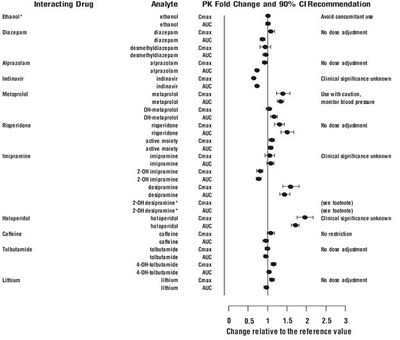
Figure 3: Pharmacokinetics of venlafaxine and its metabolite O-desmethylvenlafaxine (ODV) in special populations. - venlafaxine fig3
Venlafaxine HCl Extended-Release Capsules 75mg - venlafaxine fig4

This is a description of Venlafaxine Hydrochloride Extended-Release Capsules 75mg. It is a generic form of Effexor ER and is manufactured by Aurobindo Pharma Limited. Each capsule contains Venlafaxine hydrochloride, USP equivalent to 75mg of venlafaxine. The package size, expiration date, lot number, batch number, and other details about the product are not available. The capsule is orange in color and has the inscription £ 75 o =. The drug should only be taken as prescribed by a doctor, and it is illegal to transfer it to anyone other than the patient. The rest of the text includes instructions for taking the drug and details for billing purposes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
