Product Images Natpara (parathyroid Hormone)
View Photos of Packaging, Labels & Appearance
- Figure 1 - natpara 01
- Figure 2 - natpara 02
- Cartridge Tracker - natpara 03
- Figure A - natpara 04
- Figure B - natpara 05
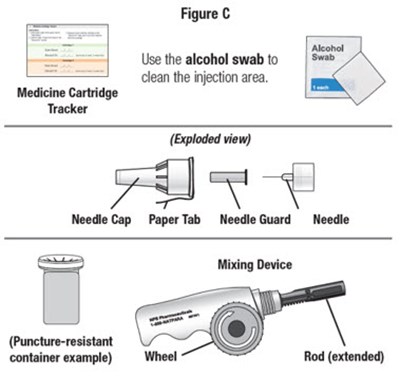
- Figure C - natpara 06
- Figure D - natpara 07
- Figure E - natpara 08
- Figure F - natpara 09
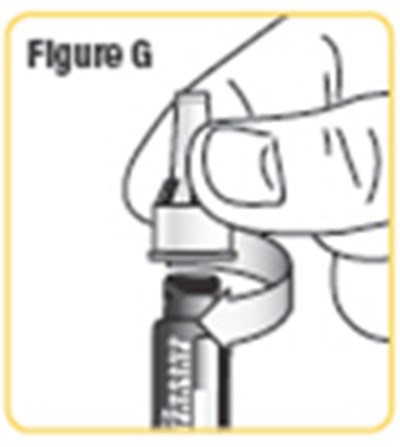
- Figure G - natpara 10
- Figure H - natpara 11
- Figure I - natpara 12
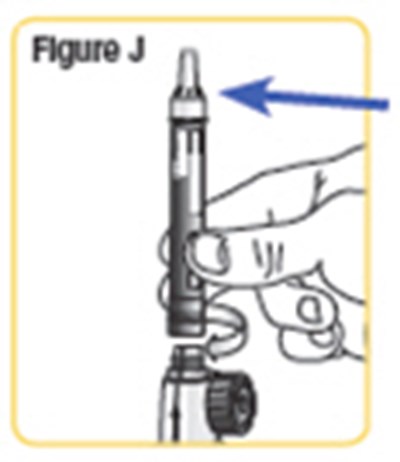
- Figure J - natpara 13
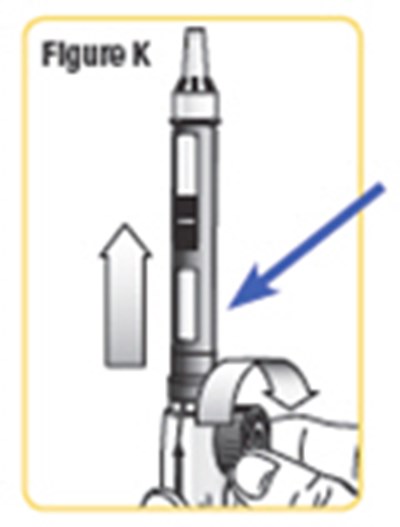
- Figure K - natpara 14
- Figure L - natpara 15
- Figure M - natpara 16
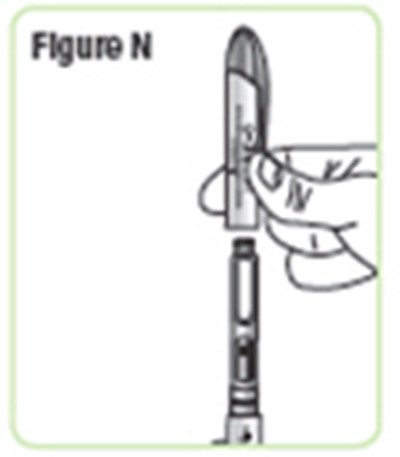
- Figure N - natpara 17
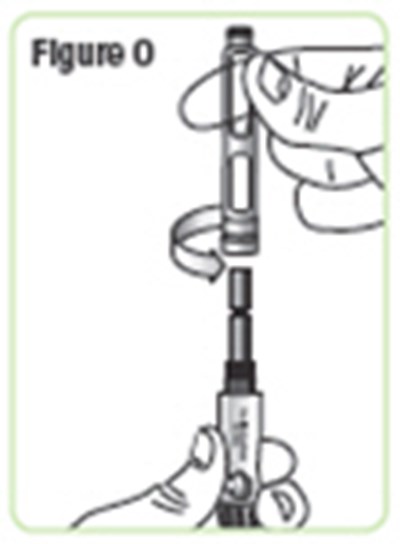
- Figure O - natpara 18
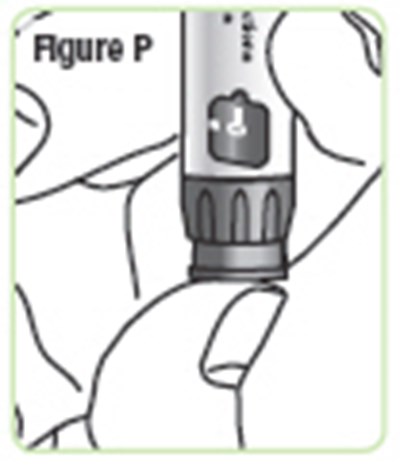
- Figure P - natpara 19
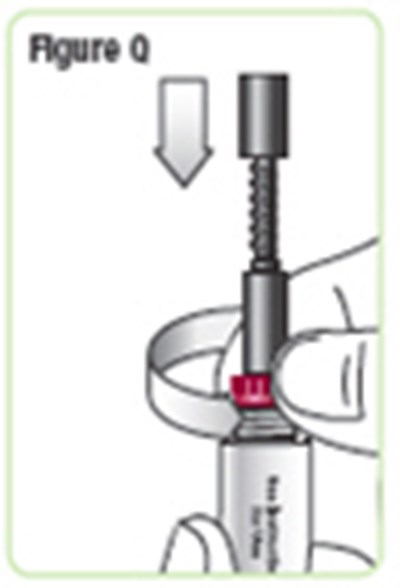
- Figure Q - natpara 20
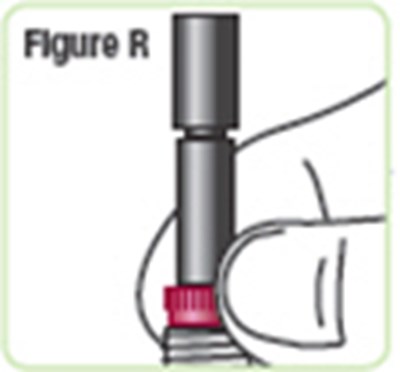
- Figure R - natpara 21
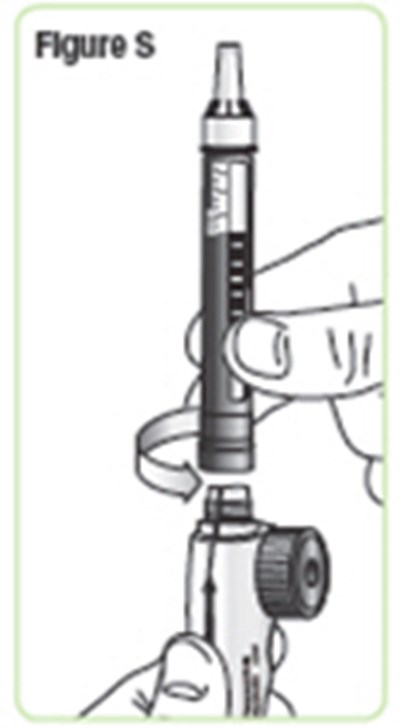
- Figure S - natpara 22
- Figure T - natpara 23
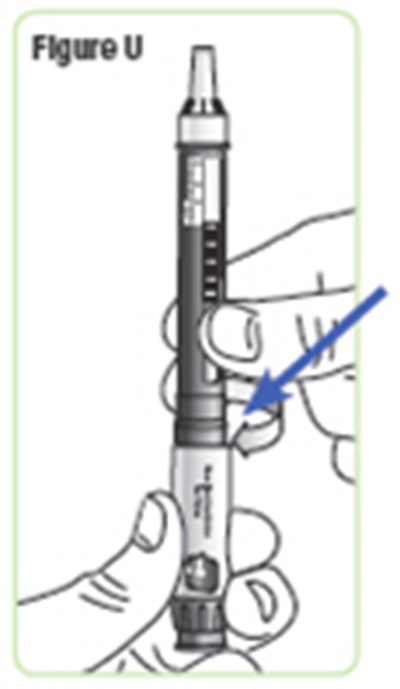
- Figure U - natpara 24
- Figure V - natpara 25
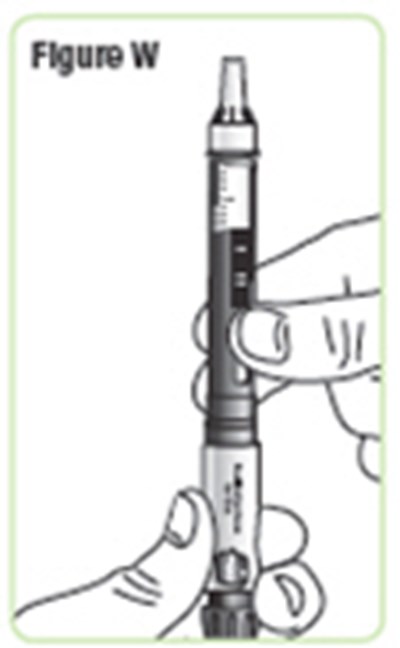
- Figure W - natpara 26
- Figure X - natpara 27
- Figure Y - natpara 28
- Figure Z - natpara 29
- Figure AA - natpara 30
- Figure BB - natpara 31
- Figure CC - natpara 32
- Figure DD - natpara 33
- Figure EE - natpara 34
- Figure FF - natpara 35
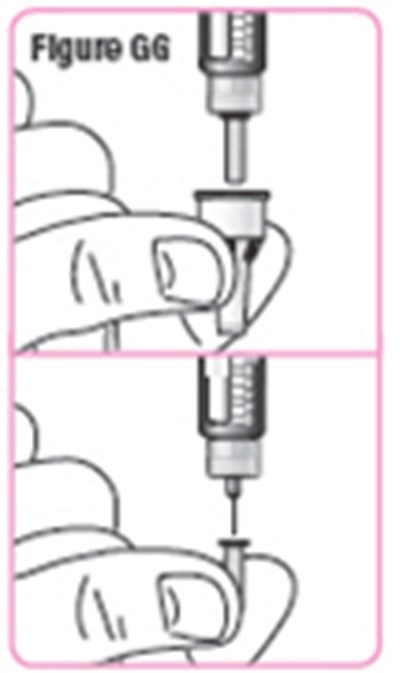
- Figure GG - natpara 36
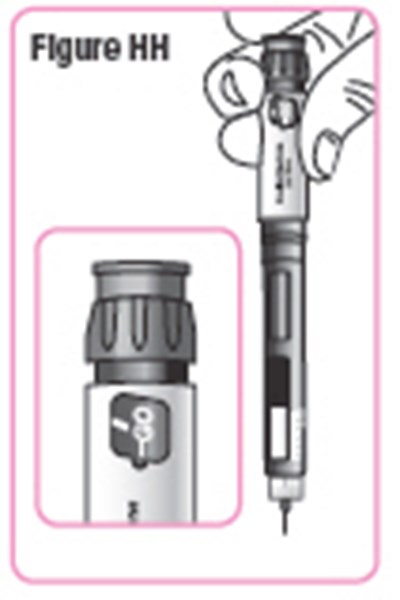
- Figure HH - natpara 37
- Figure II - natpara 38
- Figure JJ - natpara 39
- Figure - natpara 40
- Figure KK - natpara 41
- Figure LL - natpara 42
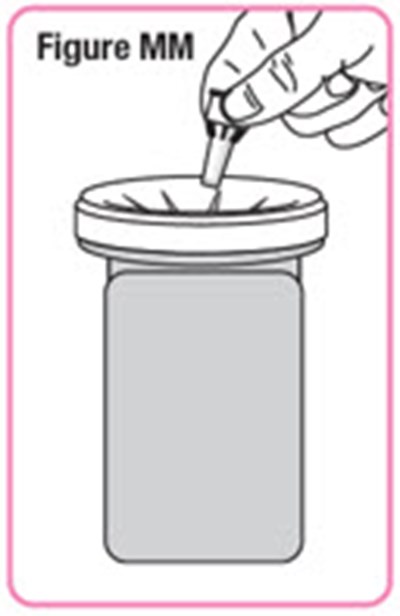
- Figure MM - natpara 43
- Figure NN - natpara 44
- PRINCIPAL DISPLAY PANEL - 25 mcg Cartridge Carton - natpara 45
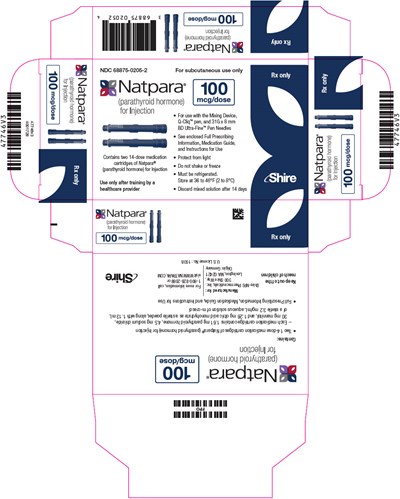
- PRINCIPAL DISPLAY PANEL - 50 mcg Cartridge Carton - natpara 46
- PRINCIPAL DISPLAY PANEL - 75 mcg Cartridge Carton - natpara 47
- PRINCIPAL DISPLAY PANEL - 100 mcg Cartridge Carton - natpara 48
Product Label Images
The following 48 images provide visual information about the product associated with Natpara (parathyroid Hormone) NDC 68875-0204 by Takeda Pharmaceuticals America, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - natpara 02
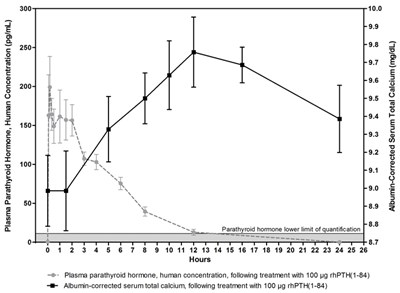
This is a graph showing the concentration of plasma parathyroid hormone (in pg/mL) following treatment with 100 g PTH (1-84). The X-axis represents hours, and the Y-axis shows the concentration of the hormone. The lower limit of quantification for parathyroid hormone is 10.0 pg/mL. Additionally, the graph also shows albumin-corrected serum total calcium (in mg/dL) levels obtained after treatment with 100 g thPTH (1-84).*
Cartridge Tracker - natpara 03

This appears to be labeling information related to cartridges and their expiration dates. The two cartridges (labeled "Cartridge 1" and "Cartridge 2") have been mixed on a certain date, and each cartridge has a specific "discard on" date. It seems that cartridge 1 has no specific date but should be simply discarded when it is used later. Cartridge 2, on the other hand, can be used until two weeks after the date it was mixed (which is not available from the given text). Overall, this seems to be an inventory management system to ensure that cartridges are used before they expire.*
PRINCIPAL DISPLAY PANEL - 25 mcg Cartridge Carton - natpara 45

This is a description of a medication called Natpara. It is a subcutaneously administered injection used for the treatment of parathyroid hormone deficiency. The medication is packaged in cartridges, each containing 14 doses, and it must be refrigerated. A healthcare provider must administer the drug, and it comes with a mixing device and needles. The medication must be used within 14 days of mixing. The text contains some gibberish, but the description manages to give information on the essential features of the medication.*
PRINCIPAL DISPLAY PANEL - 50 mcg Cartridge Carton - natpara 46
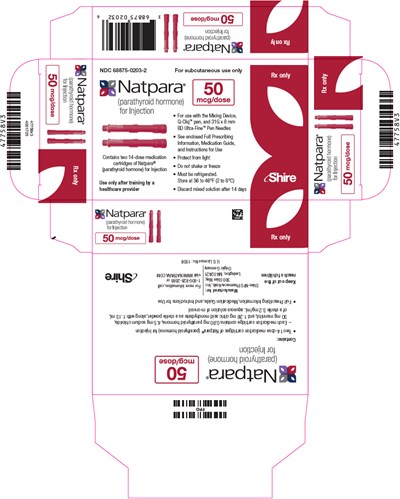
This is a description of a medication that contains parathyroid hormone, Natpara. It is designed for subcutaneous use only and comes in a packaged pen that requires BD Ultra-Fine Pen Needles. It needs to be refrigerated to be stored, and the solution should be discarded after 14 days. There are instructions for use included in this package, with a warning to not freeze and to store at a specific temperature range.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.