Product Images Panzyga
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Panzyga NDC 68982-820 by Octapharma Usa Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
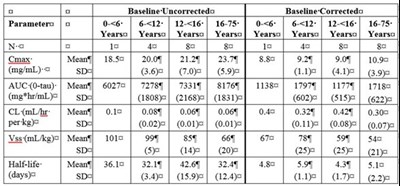
Image File - img0006

Description: This is an output of a pharmaceutical product named "Immune Globulin Intravenous (Human)", manufactured by Octapharma. The text contains some information related to the product's concentration and packaging, however, the text is not comprehensive.*
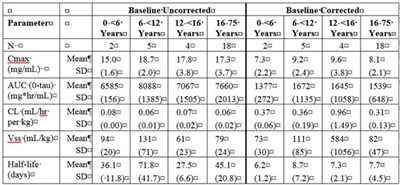
Image File - img0007

The text appears to be a list of various products that are related to immune globulin intravenous therapies, with details regarding different variants and concentrations of the products.*
Image File - img0008

This is a package for Immune Globulin Intravenous. It contains three separate products: one labeled Guman 201G, one labeled Hma 209, and one labeled Gman 1209. They are all for intravenous use, meaning they are intended to be given through a vein. All three are labeled as having been solvent and detergent-treated, and all are 200mL in volume. There is limited details available in this text.*
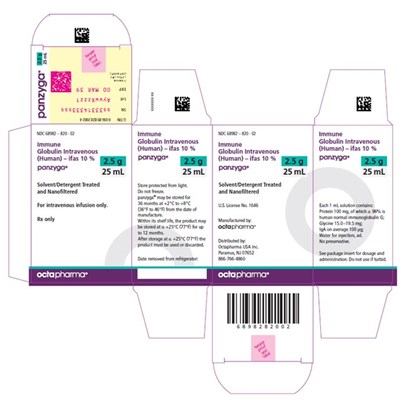
Image File - img0009

This appears to be a product description for a medication called Panzyga. It is an immune globulin intravenous solution, containing 10% Cias. The solution comes in a 300mL bottle and has been treated with a detergent. The text mentions the product in various languages, but does not provide any additional information beyond what has been stated.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.