300mcg (1) 0.5 mL Syringe Carton. (300mcg (1) 0.5 mL Syringe Carton)
 0.5 mL Syringe Carton.jpg)
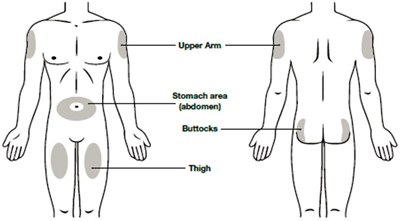
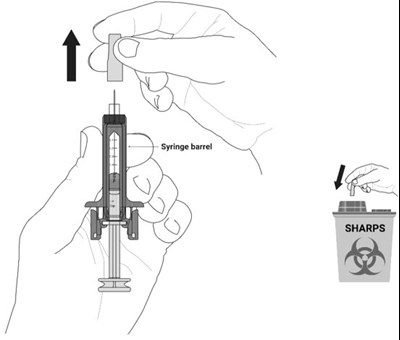
This text appears to be a description of a pharmaceutical product called NYPOZI™ Single-Dose Prefilled Syringe, which contains the medication filgrastim-txid in the form of an injection. The product is a Recombinant Granulocyte Colony Stimulating Factor derived from E. Coli, intended for subcutaneous or intravenous use. Instructions are given on how to dilute the medication for intravenous administration and under what conditions the product should be stored. The manufacturer is Tanvex BioPharma USA, Inc. from San Diego, CA. The product is indicated to be refrigerated and kept away from light. It is also mentioned to be kept out of reach of children.*
 0.5 mL Syringe Carton.jpg)
 0.8 mL Syringe Carton.jpg)
 0.8 mL Syringe Carton.jpg)