Product Images Palonosetron Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Palonosetron Hydrochloride NDC 69097-927 by Cipla Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
04460041 adec 463b ba9d 6a12038ab672 02
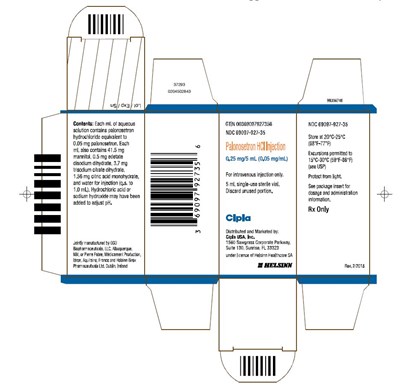
This is a product description for an intravenous injection containing palonosetron hydrochloride, used to prevent nausea and vomiting caused by chemotherapy treatment. Each milliliter of aqueous solution contains 0.05 mg of palonosetron hydrochloride, along with other ingredients. The medication is available as a single-use sterile vial, and should be discarded if not fully used. It is manufactured and distributed by Cipla USA, Inc. and dispensed only with a prescription. Storage suggestions are provided, along with instructions for use.*
04460041 adec 463b ba9d 6a12038ab672 03
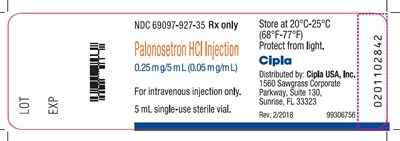
This is a description of Palonosetron HCl Injection, which is a medication that must be stored at a temperature between 20°C and 25°C (68°F-77°F). The product is produced by Cipla and comes in a 5 mL single-use sterile vial that is for intravenous injection only. The label includes a Lot number ("4 LOT"), an expiration date ("EXP"), an NDC number ("69097-927-35"), and a warning to protect it from light. The address of Cipla is provided as well.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



