Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
- PRINCIPAL DISPLAY PANEL - 100 mg - pregabalincaps 100mg 90s count
- PRINCIPAL DISPLAY PANEL - 150 mg - pregabalincaps 150mg 90s count
- PRINCIPAL DISPLAY PANEL - 200 mg - pregabalincaps 200mg 90s count
- PRINCIPAL DISPLAY PANEL - 225 mg - pregabalincaps 225mg 90s count
- PRINCIPAL DISPLAY PANEL - 25 mg - pregabalincaps 25mg 90s count
- PRINCIPAL DISPLAY PANEL - 300 mg - pregabalincaps 300mg 90s count
- PRINCIPAL DISPLAY PANEL - 50 mg - pregabalincaps 50mg 90s count
- PRINCIPAL DISPLAY PANEL - 75 mg - pregabalincaps 75mg 90s count
- Figure 1 - pregabalincaps fig01
- Figure 2 - pregabalincaps fig02
- Figure 3 - pregabalincaps fig03
- Figure 4 - pregabalincaps fig04
- Figure 5 - pregabalincaps fig05
- Figure 6 - pregabalincaps fig06
- Figure 7 - pregabalincaps fig07
- Figure 8 - pregabalincaps fig08
- Figure 9 - pregabalincaps fig09
- Figure 11 - pregabalincaps fig10
- Figure 12 - pregabalincaps fig11
- Cockcroft and Gault equation - pregabalincaps formula
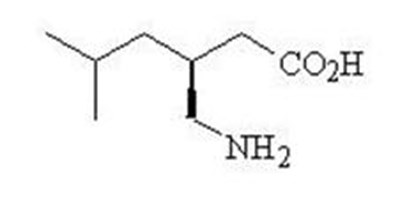
- Chemical Structure - pregabalincaps str
Product Label Images
The following 21 images provide visual information about the product associated with Pregabalin NDC 69097-954 by Cipla Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 100 mg - pregabalincaps 100mg 90s count

This is a description of a medication with the NDC code 69097-957-05 for Pregabalin, USP. Each capsule contains 100 mg of the medicine and is only for prescription use. The typical dosage is available in the package insert. The package is child-resistant and the capsules should be stored at a temperature between 20° to 25°C. The medication guide should be dispensed separately to each patient by the pharmacist. The medication is manufactured by InvaGen Pharmaceuticals, Inc., a subsidiary of Cipla Ltd., and sold under the brand Cipla in the USA. The manufacturing location is in Hauppauge, NY and it includes 90 capsules per package.*
PRINCIPAL DISPLAY PANEL - 150 mg - pregabalincaps 150mg 90s count

Each capsule of this medication contains 150 mg of Pregabalin. This package is designed to be child-resistant and should be kept out of reach of children. Store at a temperature between 20° to 25°C (68° to 77°F). Medication Guide provided by the pharmacist should be dispensed separately to each patient. This medication is manufactured by InvaGen Pharmaceuticals, Inc., a subsidiary of Cipla Ltd., in Hauppauge, NY. Cipla USA, Inc. manufactures this 90 capsule package and is located at 10 Independence Boulevard, Suite 300, Warren, NJ 07053.*
PRINCIPAL DISPLAY PANEL - 200 mg - pregabalincaps 200mg 90s count

This is a prescription medication for Pregabalin Capsules, manufactured by InvaGen Pharmaceuticals, a subsidiary of Cipla Ltd. Each capsule contains 200 mg of pregabalin, USP. The package includes 90 capsules, a medication guide, and should be stored at 20° to 25°C (68° to 77°F). The usual dosage information can be found in the package insert. This package is child-resistant and should be kept out of reach of children. Lot and expiration information are also provided.*
PRINCIPAL DISPLAY PANEL - 225 mg - pregabalincaps 225mg 90s count

The text provides details about a medication called Pregabalin which is available in capsule form manufactured by Cipla. Each capsule contains 225mg of Pregabalin, and the usual dosage is mentioned in the package insert. The medication is available in a child-resistant package and should be kept out of the reach of children. The medication guide provided should be dispensed separately to every patient. The manufacturing details of the medication are given in the text as well.*
PRINCIPAL DISPLAY PANEL - 25 mg - pregabalincaps 25mg 90s count

This is a medication guide for Pregabalin capsules. The usual dosage information can be found in the package insert. The package contains 90 capsules with 25mg of Pregabalin in each. It is child-resistant and should be kept out of reach of children. The capsules should be stored at 20° to 25°C. This medication was manufactured by InvaGen Pharmaceuticals but is sold by Cipla USA. The provided text lacks readability.*
PRINCIPAL DISPLAY PANEL - 300 mg - pregabalincaps 300mg 90s count

NDC 69097-962-05 is a prescription medicine. These capsules contain 300 mg of pregabalin, USP. It is advisable to dispense the Medication Guide separately to each patient. The manufacturer is InvaGen Pharmaceuticals, Inc., which is a subsidiary of Cipla Ltd. The usual dosage guideline can be found in the package insert. The package is child-resistant. It should be stored between 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Keep it away from children. Lot and Expiry details are provided as Rev.04/2020.*
PRINCIPAL DISPLAY PANEL - 50 mg - pregabalincaps 50mg 90s count

Pregabalin Capsules by Cipla containing 50mg of active ingredient per capsule packaged as 90 capsules per bottle. This prescription medication is dispensed with a medication guide to be given to each patient. The usual dosage is indicated in the package insert. The container is child-resistant and should be stored at 20°C to 25°C. Manufactured in NY by InvaGen Pharmaceuticals, a subsidiary of Cipla Ltd. for Cipla USA Inc. in Warren NJ. Lot number not available. This medication requires a prescription.*
PRINCIPAL DISPLAY PANEL - 75 mg - pregabalincaps 75mg 90s count

NDC 69097-956-05 Rx Only Pregabalin Capsules. Each of the 90 capsules in this child-resistant package contains 75mg of pregabalin, USP. Dispense with the medication guide provided and store at 20-25°C. Manufactured by InvaGen Pharmaceuticals, Inc. for Cipla USA, Inc. Usual dosage information can be found in the package insert.*
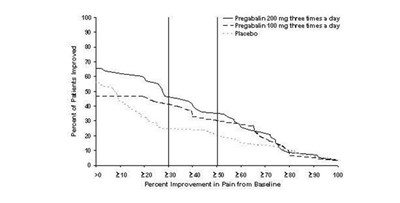
Figure 2 - pregabalincaps fig02

The text appears to be presenting data in the form of numbers under different headings like "Percent of Patients Improved" and "Percent Improvement in Pain from Baseline". The drug "Pregabalin" is mentioned along with its dosage. However, there is a random mention of "Facebook" which seems to be unrelated to the other information presented. Overall, this text seems to be missing crucial details and context, making it difficult to provide a useful description.*
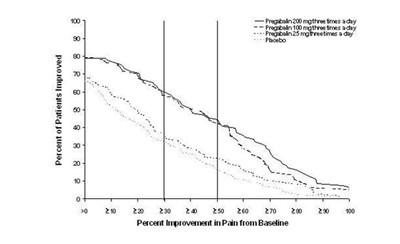
Figure 5 - pregabalincaps fig05

The text seems to describe a study of three different treatments for pain relief: Fregabalin 100mg taken three times a day, Pregataiin 80mg taken three times a day, and a placebo. The document includes a chart that may show the percentage of patients who improved with each treatment, as well as a graph showing the percent improvement in pain from baseline with each treatment. Unfortunately, the text contains several errors, making parts of it difficult to understand.*
Figure 8 - pregabalincaps fig08

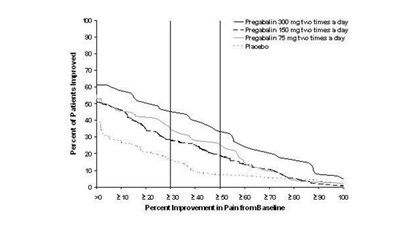
This is a graph showing the percent of patients who have improved their pain after taking different doses of Pregabalin daily. The x-axis shows the percent improvement in pain from the baseline, while the y-axis shows different doses of Pregabalin and a placebo. No further information is available.*
Figure 9 - pregabalincaps fig09

This appears to be a table showing the estimated percentage of subjects without LTR (not sure what this stands for) for a drug called "Progabain" compared to a placebo over a period of time (up to 180 days). The table shows percentages ranging from 0% to 100%, with the percentage decreasing over time for both Progabain and the placebo.*
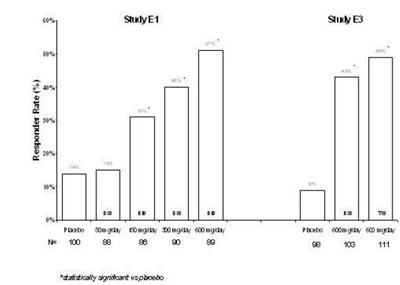
Figure 12 - pregabalincaps fig11
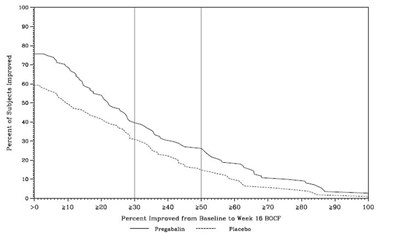
This appears to be a chart or graph with columns representing different values of improvement percentages from baseline to week 16 for two groups: one taking Pregabalin and the other taking Placebo. The x-axis shows the corresponding values for different improvements while the y-axis is not labeled. There are no units of measurement or additional context provided.*
Cockcroft and Gault equation - pregabalincaps formula
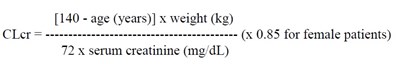
This appears to be a formula for calculating creatinine clearance (CLCT), a measure of kidney function, using the patient's age, weight, and serum creatinine level. The formula varies slightly for female patients (by multiplying the result by 0.85), but it is not clear what units the "mmmmmmmmm e e" represent.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.