Product Images Lamotrigine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 27 images provide visual information about the product associated with Lamotrigine NDC 69117-0046 by Yiling Pharmaceutical, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Proportion of Patients by Responder Rate for Lamotrigine and Placebo Group (Primary Generalized Tonic-Clonic Seizures Study) - figure 1

The text is a chart showing the responder rate, improvement, and worsening of a placebo group and a group taking lamotrigine medication. The chart displays percentages of patients in various groups. No additional context or information is available from the given text.*
Figure 2. Proportion of Patients by Responder Rate for Lamotrigine and Placebo Group (Partial-Onset Seizure Study) - figure 2
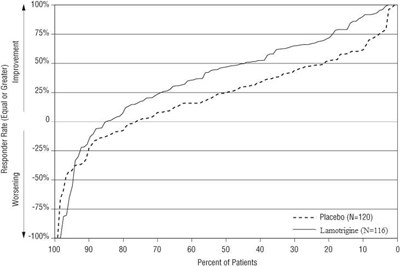
This table depicts the responder rate, improvement, and worsening percentages for patients who were given lamotrigine or a placebo. The data is broken down by a scale of 4, with a 100% responder rate for those who had an equal or greater improvement as well as a 5% worsening rate in the lamotrigine group. The placebo group saw a 50% worsening rate. The table includes numbers and percentages of patients who fall under these categories, in total, 120 were given a placebo and 116 were given lamotrigine.*
25mg 30tablets - label 01

This is a prescription drug called Once A Day Lamotrigine, with a dosage of 25mg per tablet. It is manufactured by Yiling Pharmaceutical Ltd based in Hebei, China and distributed by Yiling Pharmaceutical Inc. in Las Vegas, USA. The drug should be kept at room temperature between 20°C to 25°C with excursions allowed between 15°C to 30°C. The batch number is available, and it is advised not to use the drug if the safety seal under the cap is broken or missing. Further dosage information can be obtained from the prescribing information guide.*
25mg 100tablets - label 02

This appears to be the label of medication manufactured by Yiling Pharmaceutical Ltd. The medication is called Once A Day Lamotrigine, USP 25 mg and each tablet contains 100mm by 40mm. The label specifies the manufacturer's details, distributor, batch number, and expiry date. The label also cautions users to verify the product and not to use it if the safety seal is broken or missing. The medication guide for the product is also mentioned.*
25mg 500tablets - label 03

This is a description of a medication package containing 500 tablets of Lamotrigine, USP 25mg, manufactured by Yiling Pharmaceutical Ltd in China and distributed by Yiling Pharmaceutical, Inc. in the USA. The package includes a caution to verify the product dispensed and to dispense the accompanying medication guide to each patient. The tablets are to be taken once a day and dosage information is available in the prescribing information. The package also includes a warning not to use if the printed safety seal under the cap is broken or missing and instructions to store the medication at temperatures between 20°C to 25°C (68°F to 77°F) with permitted excursions to 15°C to 30°C (59°F to 86°F).*
50mg 100tablets - label 05

These are details about a pharmaceutical product manufactured by Yiling Pharmaceutical Ltd. The tablets are extended-release Lamotrigine, USP of strength 50mg. The bottle contains 100 tablets and comes with a safety seal under the cap. The product should be stored between 20 to 25°C and if the seal is broken or missing it should not be used. The batch number, expiry date, manufacturer, and distributor's details are also provided. The product is for prescription use only and comes with a medication guide to be given to each patient.*
50mg 500tablets - label 06

This is a description of a medication that comes in tablet form. The tablets are 80mm and 180mm and the box contains 500 tablets. They are manufactured by Yiling Pharmaceutical Ltd in China and distributed by Yiling Pharmaceutical, Inc. in the USA. The medication contains Lamotrigine, USP 50 mg and is an extended-release tablet designed to be taken once a day. There is a caution to verify the product dispensed and to dispense the accompanying medication guide to each patient. The product should be stored between 20°C to 25°C, with excursions permitted to 15°C to 30°C. The batch number, expiry date, GTIN, and Rev number are also included.*
100mg 30tablets - label 07

YILING Lamotrigine, USP 100 mg is an extended-release tablet produced by Yiling Pharmaceutical Ltd in China, distributed in the USA by Yiling Pharmaceutical, Inc. Each tablet contains exactly 100mm, with a batch number, expiry date, and prescription warning to ensure product safety. A medication guide must be dispensed with each prescription. Store in room temperature conditions. Detailed prescribing information is available.*
100mg 100tablets - label 08

This is a description for Lamotrigine, USP 100mg extended-release tablets. The tablets are distributed by Yiling Pharmaceutical, Inc. in the USA and manufactured by Yiling Pharmaceutical Ltd in China. Each bottle contains 100 tablets with a caution to verify the product dispensed. A medication guide is dispensed with each tablet. The expiry date and batch number are available, and the product should be stored at a temperature between 20-25°C.*
100mg 500tablets - label 09

This is a description of a medication package containing 500 tablets of 100mg Lamotrigine extended-release USP tablets. The tablets are produced by Yiling Pharmaceutical Ltd in China and distributed in the USA by Yiling Pharmaceutical, Inc. The package includes a medication guide for patients, and caution must be taken to verify the product dispensed. The dosage information is included in the prescribing information. The tablets should be stored at a controlled room temperature of 20-25°C (68-77°F).*
200mg 100tablets - label 11

This is a description of a medication identified as "YILING Lamotrigine, USP 200 mg". The medication comes in an extended-release tablet form with a dosage information printed in the prescribing information. The medication is meant for once-a-day use and is supplied in a bottle containing 100 tablets. It is manufactured by Yiling Pharmaceutical Ltd located in Hebei, China, and distributed by Yiling Pharmaceutical, Inc. located in Las Vegas, USA. The label contains information about the caution to verify the product dispensed, the need to dispense a medication guide to each patient, and storage instructions. The text also includes a product code (NDC 69117-0047-2) and batch numbers.*
200mg 500tablets - label 12

This is a description of a medication manufactured by Yiling Pharmaceutical Ltd that contains Lamotrigine, USP 200 mg. Its distributor is Yiling Pharmaceutical, Inc. and it comes in a bottle of 500 tablets with batch number and expiry date printed. It is an extended-release tablet used for once-a-day dosing. A medication guide should be given to each patient as caution. Its usage and dosage information are included in the prescribing information. Store it at a temperature between 20°C to 25°C (68°F to 77°F).*
250mg 30tablets - label 13

The text is a product label for medication containing Lamotrigine, USP 250 mg manufactured by Yiling Pharmaceutical Ltd. The tablets are extended-release and should be taken once a day. The safety seal should not be broken or missing. The medication guide should be provided to each patient. The storage temperature should be between 20° to 25°C (68° to 77°F) with excursions permitted to 15° t0 30°C (59° to 86°F). The batch number, expiry date, GTIN, LOT, and EXP are not available.*
250mg 100tablets - label 14

YILING pharmaceuticals is a US-based distributor of lamotrigine tablets manufactured by Yiling Pharmaceutical Ltd in China. The tablets are extended-release and contain 250mg of Lamotrigine, USP. Each bottle contains 100 tablets, and the recommended dosage should be confirmed by referring to the prescribing information. They are to be stored within a temperature range of 20°C to 25°C (68°F to 77°F), with excursions permitted to 15°F to 30°C (59°F to 86°F). Patients must be given a medication guide before use.*
250mg 500tablets - label 15

This is a label of tablets containing Lamotrigine, USP 250 mg manufactured by Yiling Pharmaceutical Ltd. It includes important dosage and usage instructions and should be stored at room temperature. The bottle contains 500 tablets and is sealed for safety purposes.*
300mg 30tablets - label 16

This is a product label for a package of 30 tablets of Once A Day Lamotrigine, USP 300mg extended-release tablets, manufactured by Viling Pharmaceuticals in Hebei, China, and distributed by Yiling Pharmaceutical, Inc. in Las Vegas, NV, USA. The label includes information such as the batch number, expiry date, and caution to verify the product dispensed. Storage instructions and a safety seal warning are also given.*
300mg 100tablets - label 17
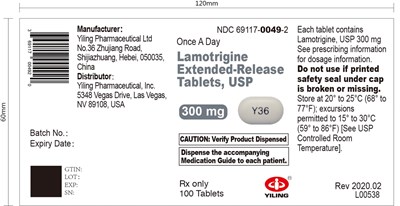
Yiling Pharmaceutical's Lamotrigine Extended-Release Tablets, USP are available in a batch of 100, with each tablet containing 300mg of Lamotrigine. The distributor of this medication is Yiling Pharmaceutical, Inc. in Las Vegas, NV, USA. The medication guide must be dispensed to each patient. The manufacturer is located in Hebei, China, and their address is included in the description. The expiry date, as well as NDC number, is not available in the provided text.*
300mg 500tablets - label 18

This is a medication called Lamotrigine, manufactured by Yiling Pharmaceutical Ltd in China. The medication comes in the form of 130mm extended-release tablets, with a dosage of 300mg of Lamotrigine per tablet. There are 500 tablets in total in the package. The medication guide should be dispensed to each patient along with the product. There is a caution to verify the product dispensed and the tablets should not be used if the printed safety seal under the cap is broken or missing. The tablets should be stored at 20° to 25°C (68° to 77°F), with excursions permitted to 15° 0 30°C (59° to 86°F) [See USP Controlled Room Temperature].*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.