Product Images Dexmethylphenidate Hydrochloride Extended-release
View Photos of Packaging, Labels & Appearance
- Dexmethylphenidate XR 20 mg capsule Label - 6918926851
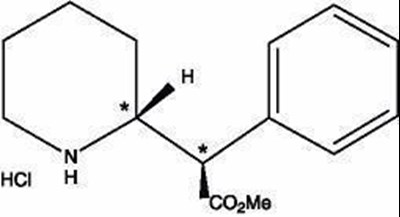
- Dexmethylphenidate hydrochloride structural formula. - abd467a8 0340 4f41 9eed e7701b06b114 01
- Figure 1 Mean Dexmethylphenidate Plasma Concentration-Time Profiles After Administration of 1 x 20 mg Dexmethylphenidate Hydrochloride Extended-Release Capsules (n=24) and 2 x 10 mg Dexmethylphenidate Hydrochloride Immediate-Release Tablets (n=25) - abd467a8 0340 4f41 9eed e7701b06b114 02
Product Label Images
The following 3 images provide visual information about the product associated with Dexmethylphenidate Hydrochloride Extended-release NDC 69189-2685 by Avera Mckennan Hospital, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Dexmethylphenidate XR 20 mg capsule Label - 6918926851

This is a description of a medication called Dexmethylphenidate XR manufactured by Sandoz. It is a 20 mg capsule with a manufacturing lot number of F0004 and an expiration date of 11/30/2016. The National Drug Code (NDC) for this medication is 691 8926851. It appears to have been intended for distribution to a hospital or medical facility for use in Class C-1 patients. The remaining text after the NDC number is not readable.*
Figure 1 Mean Dexmethylphenidate Plasma Concentration-Time Profiles After Administration of 1 x 20 mg Dexmethylphenidate Hydrochloride Extended-Release Capsules (n=24) and 2 x 10 mg Dexmethylphenidate Hydrochloride Immediate-Release Tablets (n=25) - abd467a8 0340 4f41 9eed e7701b06b114 02
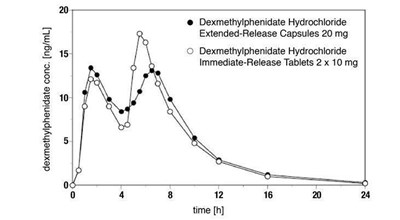
This is a table showing the concentration of dexmethylphenidate in ng/mL with values of 20 and 15. The table also mentions Dexmethylphenidate Hydrochloride Extended-Release Capsules 20 mg and Immediate-Release Tablets 2 x 10 mg.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.