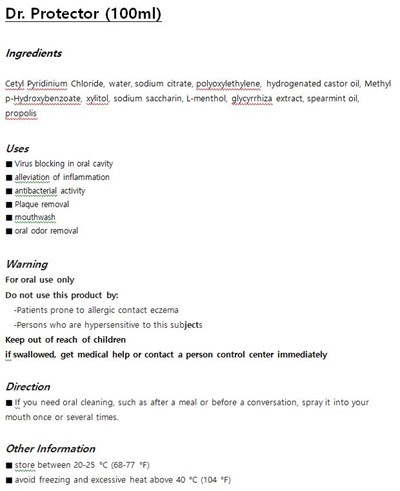
FDA Label for Dr. Protector
View Indications, Usage & Precautions
Dr. Protector Product Label
The following document was submitted to the FDA by the labeler of this product Sungwon Cosmetic Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Cetyl Pyridinium Chloride
Inactive Ingredient
water, sodium citrate, etc
Otc - Purpose
for dental care
Otc - Keep Out Of Reach Of Children
KEEP OUT OF REACH OF THE CHILDREN
Indications & Usage
If necessary, spray several times orally
Warnings
- Do not use if mouth is loose or eczema
-.Storage and handling precautions
1)If possible, avoid direct sunlight and store in cool and area of low humidity
2)In order to maintain the quality of the product and avoid misuse
3)Avoid placing the product near fire and store out in reach of children
Dosage & Administration
for oral use only
Package Label.Principal Display Panel
* Please review the disclaimer below.