Product Images Tiagabine Hydrochloride
View Photos of Packaging, Labels & Appearance
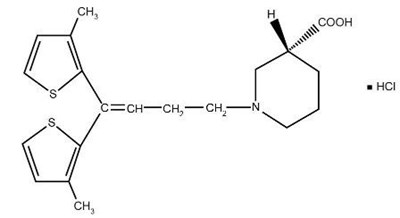
- chemical structure - tiagabine hydrochloride tablets 1
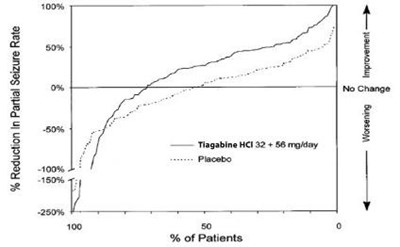
- fig1 - tiagabine hydrochloride tablets 2
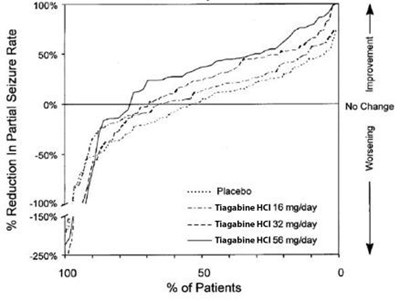
- fig2 - tiagabine hydrochloride tablets 3
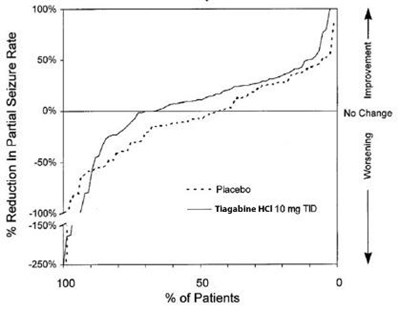
- fig3 - tiagabine hydrochloride tablets 4
- fig4 - tiagabine hydrochloride tablets 5
- 1 - tiagabine hydrochloride tablets 6
- 2 - tiagabine hydrochloride tablets 7
- 3 - tiagabine hydrochloride tablets 8
- 1 - tiagabine hydrochloride tablets 9
Product Label Images
The following 9 images provide visual information about the product associated with Tiagabine Hydrochloride NDC 69238-1106 by Amneal Pharmaceuticals Ny Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
fig3 - tiagabine hydrochloride tablets 4
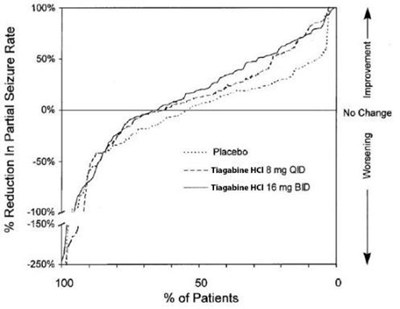
The text provides a chart for the "Reduction in Partial Seizure Rate" with the percentage of patients on placebo and different doses of Tiagabine. The chart shows that Tiagabine at 8mg once daily (QD) resulted in a 50% reduction in partial seizure rate, while Tiagabine at 16mg twice daily (BD) resulted in a 100% reduction in partial seizure rate.*
1 - tiagabine hydrochloride tablets 6

This is a medication guide for Tiagabine Hydrochloride, available in a package of 30 tablets. The medication guide can be obtained by scanning the provided QR code or visiting a specific website. The guide should be dispensed to every patient by the pharmacist.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.