Product Images Clonidine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Clonidine Hydrochloride NDC 69238-1426 by Amneal Pharmaceuticals Ny Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - clonidine hydrochloride extended release tablets 3
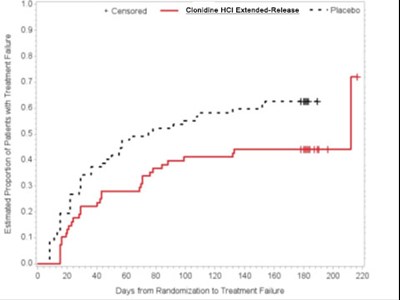
Estimated Proportion of Patients with Treatment Failure over time is plotted in the given graph. The X-axis represents the Days from Randomization to Treatment Failure and the Y-axis represents the percentage (10% to 100%). The graph seems to show an increasing trend in the proportion of patients with treatment failure over time. However, due to the limited information provided, it is unclear what the numbers on the graph represent.*
0.1mg 60ct Label - clonidine hydrochloride extended release tablets 4

NDC 69238-1426-6 is an extended-release tablet of Clonidine HCI, used for medicinal purposes. Each tablet contains 0.1 mg clonidine HCI and is only available through prescription. This drug should not be substituted for other clonidine products and must be kept out of reach of children. It is essential to swallow the tablet whole and not crush, cut, or chew it. It is advised to store the medication at controlled room temperature. The medication is distributed by Amneal Pharmaceuticals LLC from Bridgewater, NJ.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

