Product Images Paclitaxel
View Photos of Packaging, Labels & Appearance
- 100mg - 100 mg label
- 100mg Carton - 100mg canton
- 300mg carton - 300mg carton
- 300mg - 300mg label
- 30mg carton - 30mg carton
- 30mg - 30mg label
- Figure-1 - fig1
- Figure 2 - fig2
- Figure 3 - fig3
- Figure-4 - fig4
- Figure 5 - fig5
- Figure-6 - fig6
- Figure-7 - fig7
- Figure-8 - fig8
- Structure - structure
- Table 1 - tab1
- Table 10 - tab10
- Table 11 - tab11
- Table 12 - tab12
- Table 13 - tab13
- Table 14 - tab14
- Table 15 - tab15
- Table 16 - tab16
- Table 17 - tab17
- Table 2A - tab2a
- Table 2B - tab2b
- Table 3 - tab3
- Tabel 4 - tab4
- Table 5 - tab5
- Table 6 - tab6
- Table 7 - tab7
- Table 8 - tab8
- Table 9 - tab9
Product Label Images
The following 33 images provide visual information about the product associated with Paclitaxel NDC 69339-228 by Natco Pharma Usa Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
100mg - 100 mg label

This text provides information about a medication called Paclitaxel Injection, USP. It contains 6 mg of the active ingredient and is manufactured by NATCO PHARMA LIMITED. The usual dosage instructions and precautions for handling the medication are mentioned. The text also includes details of the NDC number, storage instructions, manufacturer details, and warnings about the cytotoxic nature of the medication.*
300mg carton - 300mg carton

This text appears to be a description of a pharmaceutical product, possibly a cytotoxic agent in a multiple-dose vial. It includes information on the active ingredients, storage instructions, manufacturer details, and dosage guidelines. The text emphasizes the need for caution and proper handling due to the cytotoxic nature of the agent. It is essential to dilute the solution correctly and follow the enclosed package insert for further information.*
300mg - 300mg label

This is a medication label containing information about a Paclitaxel Injection, USP. It includes details such as the active ingredient, inactive ingredients, dosage, storage instructions, warnings, manufacturer details, and precautions. The medication is a cytotoxic agent and should be stored at specific temperature ranges and protected from light. The label also emphasizes the need for caution and recommends using gloves while handling the containers. The document further mentions the importance of referencing the package insert for detailed dosage and usage directions.*
30mg carton - 30mg carton

This text appears to be a description related to pharmaceutical products. It provides details about a semisynthetic Paclitaxel Injection, USP, which is categorized as a cytotoxic agent and requires dilution as per the enclosed package insert. It includes information about the active and inactive ingredients, storage instructions, warnings, proper dosage, and usage precautions. The text emphasizes the necessity of dilution and careful handling of the containers. It overall serves as a guide for the safe and effective use of the medication.*
Figure-1 - fig1
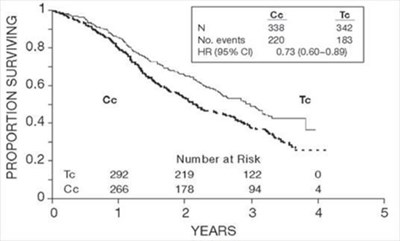
This text seems to provide data on survival rates over time, as indicated by the table showing the proportion surviving at different time points. The numbers represent the number of participants at risk at various time intervals, along with the number of events and hazard ratio with confidence intervals. This data may be used for survival analysis in a research study or clinical trial.*
Figure 2 - fig2

This text provides summarized survival data, including the number of individuals at risk over different time points and the number of events that occurred at each time point. It also presents the hazard ratio (HR) with a 95% confidence interval for the observed data. The survival pattern over time can be understood by observing the proportion of individuals surviving at each time point.*
Figure 3 - fig3
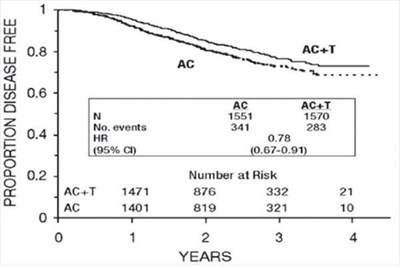
The text contains statistical data related to disease-free proportions over a period of years. This includes the number of individuals at risk, the number of events recorded, and the hazard ratio (HR) which indicates the likelihood of an event happening in one group compared to another. The confidence interval (95% CI) for the hazard ratio is also provided in the text. The data seems to be organized by different groups (AC, AC+T) and the number of individuals in each group at different time points.*
Figure-4 - fig4

This text provides information on survival proportions for different treatments over a certain period. It includes the number of patients at risk for each treatment group at various time points and the number of events observed. The hazard ratio (HR) is also given, along with confidence intervals. The data seems to extend up to four years.*
Figure 5 - fig5

Proportion of disease-free individuals in a study is presented in the format of a table. The table shows the number of events, hazard ratio, and confidence intervals. It also includes the number at risk for two different groups (AC+T and AC) over a period of 4 years. This data helps in understanding and comparing disease-free status between the two groups.*
Figure-6 - fig6

This text provides data on the proportion of individuals free of a certain disease over time. It includes details such as the number of events, hazard ratio, confidence interval, and the number at risk in different treatment groups over a span of years. The analysis suggests that the intervention (AC+T) has a hazard ratio of 0.92 with a 95% confidence interval of 0.73-1.16.*
Figure-8 - fig8

Proportion Disease Free: The text provides data on disease-free status over time, with the number of events, hazard ratio, and confidence interval. This information is crucial for evaluating the effectiveness of a treatment or intervention in maintaining a disease-free state over a specified period.*
Table 1 - tab1
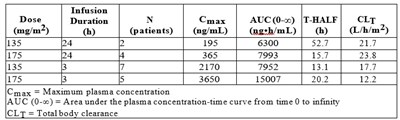
This is information on dose and infusion duration of a medication. It includes data such as Cmax (Maximum plasma concentration), AUC(0-∞) (Area under the plasma concentration-time curve from time 0 to infinity), T-HALF (Half-life), and CLT (Total body clearance) for different doses administered to patients. The information provided can be used for pharmacokinetic evaluation and to understand the drug's behavior in the body.*
Table 10 - tab10

This text provides detailed information on the percentage of patients experiencing various effects during a medical study, such as bone marrow conditions, cardiovascular parameters, peripheral neuropathy, musculoskeletal symptoms, gastrointestinal issues, and hepatic abnormalities. The study also demonstrates the significance of premedication and outlines the criteria for defining severe events. The data is presented based on the worst course analysis.*
Table 11 - tab11
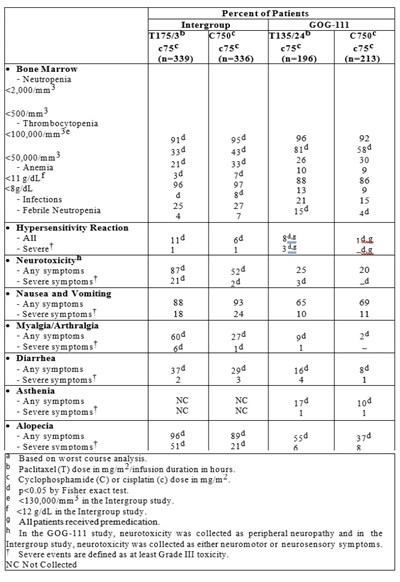
This text seems to provide detailed statistical data related to the Intergroup GOG 11 study, including information on the percentage of patients experiencing various symptoms, side effects, and toxicities during the treatment. The data covers aspects such as bone marrow health, neutropenia, thrombocytopenia, anemia, infections, hypersensitivity, neurotoxicity, nausea, vomiting, myalgia/arthralgia, diarrhea, asthenia, and alopecia. The document also includes information on specific medications (Paclitaxel and Cyclophosphamide/Cisplatin), dosage details, statistical significance (p-value), and criteria for defining severe events. Additionally, it provides notes on data collection methodologies and parameters used in the study. Overall, this text is valuable for evaluating the impact and outcomes of the treatment administered in the study.*
Table 12 - tab12

This is a statistical analysis of patient outcomes in a clinical study examining the effects of Paclitaxel treatment on various conditions such as neutropenia, thrombocytopenia, anemia, infections, hypersensitivity reactions, peripheral neuropathy, and mucositis. The data includes the percentage of patients experiencing different levels of severity for each condition. Severe events are defined as at least Grade 111 toxicity, and all patients received premedication. The study also evaluates the Paclitaxel dose in mg/m^2 and infusion duration in hours.*
Table 13 - tab13
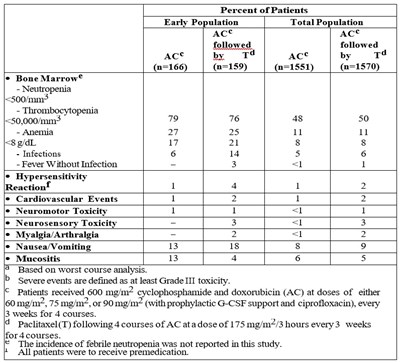
This is an evaluation of the percentage of patients experiencing different adverse events during cancer treatment with drugs such as cyclophosphamide, doxorubicin, and paclitaxel. Information on events like neutropenia, thrombocytopenia, anemia, infections, hypersensitivity reactions, cardiovascular events, and more are provided for both the early and total patient populations. The study also mentions the doses of the drugs administered to the patients and the premedication they received.*
Table 14 - tab14

This text appears to show data related to the percent of patients experiencing different medical conditions or symptoms during treatment with Paclitaxel. The data includes information on neutropenia, anemia, infections, peripheral neuropathy, and mucositis among other conditions. Severe events are defined as at least Grade III Toxicity. The patients received premedication, and the analysis seems to be based on the worst course of the treatment.*
Table 15 - tab15

This text provides information on the percent of patients experiencing different side effects and symptoms during a medical treatment regimen. It includes details on bone marrow-related issues, infections, hypersensitivity reactions, arthralgia/myalgia, nausea/vomiting, mucositis, neurotoxicity, and cardiovascular events. Severe events are classified as Grade III Toxicity. The treatment regimen involves the administration of Paclitaxel (T), cisplatin (C), and Etoposide (VP) with G-CSF support and premedication. The data is presented in a tabular format for better analysis.*
Table 17 - tab17

This text presents guidelines for adjusting the dose of Paclitaxel based on the degree of hepatic impairment, as determined by transaminase and bilirubin levels. Different dosage recommendations are provided for 24-hour and 3-hour infusions, with specific thresholds for bilirubin levels determining the appropriate dose adjustments. The recommendations highlight the importance of individual tolerance for further dose reductions in subsequent courses of therapy. Note that these guidelines are specifically for patients without hepatic impairment and may not apply to all situations, such as for AIDS-related Kaposi's sarcoma.*
Table 2A - tab2a
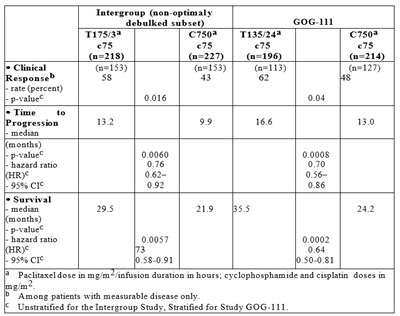
This is an evaluation of the outcomes in the non-optimally debulked subset of patients in the Intergroup GOG-111 study. The text includes data on response rates, time to progression, survival rates, and hazard ratios for different treatments. The study involves the administration of paclitaxel, doxorubicin, cisplatin, and cyclophosphamide. The outcomes are stratified for Study GOG-111.*
Table 2B - tab2b

This text appears to be a statistical report detailing clinical responses, time to progression, and survival outcomes of a certain treatment regimen. It includes information such as response rates, p-values, hazard ratios, and confidence intervals. The text also mentions specific doses of medications administered and notes that the data may only apply to patients with measurable disease. This report seems to focus on the efficacy and impact of the treatment on patients involved in the study.*
Table 3 - tab3

This text appears to contain statistical information related to rates, confidence intervals, time to progression, and survival data including medians and corresponding confidence intervals. The text presents values such as rates (percent), median time (months), and confidence intervals for each category, which may be used in a research or analytical context to evaluate outcomes or trends.*
Tabel 4 - tab4

This text provides information on Disease-Free Survival and Overall Survival rates based on different subsets of patients. It evaluates factors like the number of positive nodes, tumor size, menopausal status, and receptor status. The data includes hazard ratios with corresponding confidence intervals for various categories within each subset of patients.*
Table 5 - tab5

This text provides data from a study or clinical trial, including response rate, time to progression, and survival outcomes in terms of median months and corresponding p-values. The response rate was 28%, with a p-value of 0.135. Time to progression had a median of 42 months, a p-value of 0.027, and a value of 3.0. Survival had a median of 117 months with a p-value of 0.321, and a separate value of 10.5. These metrics can be used to evaluate the effectiveness and outcomes of the treatment or intervention being studied.*
Table 6 - tab6

This data shows the comparison of outcomes between the treatment groups T135/24 and T25024 with different parameters like response rate, time to progression, and survival rates. It includes information on the administration of Etoposide (VP) 100 mg/m² compared to cisplatin/etoposide. The study reveals differences in response rates, median time to progression, and survival rates among the groups studied.*
Table 7 - tab7

This text appears to be a clinical notation related to systemic therapy and different types of edema. It seems to categorize different conditions possibly based on their severity or specific characteristics. It mentions varying numbers associated with visceral, edema, oral, and cutaneous conditions. The text also highlights the importance of distinguishing different types of edema.*
Table 8 - tab8

This description seems to be related to the evaluation of systemic therapy outcomes based on responses such as complete response, partial response, stable disease, progression, and early death/toxicity. These terms are likely used to track the effectiveness and adverse events associated with treatment.*
Table 9 - tab9

This is a clinical study data showing the incidence of Neutropenia and Peripheral Neuropathy in patients with different types of cancer and different age groups. The study includes information on ovarian cancer, breast cancer, and non-small cell lung cancer. It also provides details on the dosage of Paclitaxel and cisplatin used in the treatments. The study categorizes Peripheral Neuropathy as neurosensory toxicity in the different cancer studies.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




