FDA Label for Flexxsonic Pain Relief
View Indications, Usage & Precautions
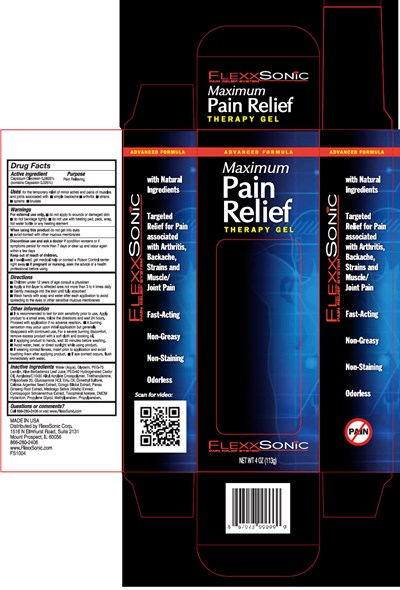
Flexxsonic Pain Relief Product Label
The following document was submitted to the FDA by the labeler of this product Flexxsonic Corporation. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Other
Drug Facts
Distributed by FlexxSonic Corp.
1516 N Elmhurst Road, Suite 2131
Mount Prospect, IL 60056
Active Ingredient
Capsicum Oleoresin 0.0625% (contains Capsaicin 0.025%)
Purpose
Pain Relieving
Uses
for the temporary relief of minor aches and pains of muscles and joints associated with
- simple backache
- arthritis
- strains
- sprains
- bruises
Warnings
For external use only.
- do not apply to wounds or damaged skin
- do not bandage tightly
- do not use with heating pad, pack, wrap, hot water bottle or any heating element When using this product do not get into eyes
- avoid contact with other mucous membranes Discontinue use and ask a doctor if condition worsens or if symptoms persist for more than 7 days or clear up and occur again within a few days
Otc - Keep Out Of Reach Of Children
Keep out of reach of children.
- if swallowed, get medical help or contact a Poison Control center right away
- if pregnant or nursing, seek the advice of a health professional before using
Directions
- Children under 12 years of age consult a physician
- Apply a thin layer to affected area not more than 3 to 4 times daily
- Gently massage into the skin until fully absorbed
- Wash hands with soap and water after each application to avoid spreading to the eyes or other sensitive mucous membranes
Other Information
- It is recommended to test for skin sensitivity prior to use. Apply product to a small area, follow the directions and wait 24 hours. Proceed with application if no adverse reaction.
- A burning sensation may occur upon initial application but generally disappears with continued use. For a severe burning discomfort, remove excess product with a soft cloth and cooking oil.
- If applying product to hands, wait 30 minutes before washing.
- Avoid water, heat, or direct sunlight while using product.
- If wearing contact lenses, insert prior to application and avoid touching them after applying product.
- If eye contact occurs, flush immediately with water.
Inactive Ingredients
Water (Aqua), Glycerin, PEG-75 Lanolin, Aloe Barbadensis Leaf Juice, PEG-40 Hydrogenated Castor Oil, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Triethanolamine, Polysorbate 20, Glucosamine HCI, Emu Oil, Dimethyl Sulfone, Celosia Argentea Seed Extract, Ginkgo Biloba Extract, Panax Ginseng Root Extract, Medicago Saliva (Alfalfa) Extract, Cymbopogon Schoenanthus Extract, Tocopheryl Acetate, DMDM Hydantoin, Propylene Glycol, Methylparaben, Propylparaben.
Questions Or Comments?
Call 866-280-2406 or visit www.FlexxSonic.com
Principal Display Panel - 113 G Bottle Carton
ADVANCED FORMULA
Maximum
Pain
Relief
THERAPY GEL
FLEXXSONiC
PAIN RELIEF SYSTEM
NET WT 4 OZ (113g)
* Please review the disclaimer below.