Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Gabapentin NDC 69434-044 by Zhejiang Yongtai Pharmaceutical Co., Ltd, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
100 mg - 100mg 100s 07

This is a description of storage guidelines for gabapentin capsules manufactured in China. The capsules contain 100 mg of gabapentin and are to be stored in temperatures ranging from 20°C to 25°C, with excursions permitted between 15°C to 30°C. Dispensing of these capsules should be done in tight, child-resistant containers. The NDC code for this medication is 69434-042-04. The manufacturer is Zhejiang Yongtai Pharmaceutical Co., Ltd. Patients are advised to consult the package insert for full prescribing information, and a medication guide should be provided to each patient.*
300 mg - 300mg 100s 08

This is a description related to medication packaging for gabapentin capsules containing 300mg with storage instructions to maintain temperature between 20 to 25°C (68°F to 77°F) and with excursions permitted between 15°C to 30°C (59°F to 86°F). Dispensing should be done in tight (USP), Capsules, USP child-resistant containers. The medication guide enclosed should be dispensed to each patient along with the package insert that contains complete prescribing information. The manufacturer is Zhejiang Yongtai Pharmaceutical Co., Ltd located in Linhai, Zhejiang, China. The information also includes the drug's National Drug Code (NDC) and the lot number.*
400 mg - 400mg 100s 09

This is a prescription medication called Gabapentin, made in China by Zhejiang Yongtai Pharmaceutical Co., Ltd. Each capsule contains 400mg of Gabapentin and should be stored in child-resistant containers between 15°C to 30°C (59°F to 86°F). The medication guide should be given to each patient. For more information on dosage and usage, please refer to the package insert. The NDC code for this medication is 69434-044-04.*
image description - gaba 01
This text provides formulas for estimating the creatinine clearance (CLCr) of a patient based on their age and weight, as well as their serum creatinine level. It also includes a correction factor for female patients.*
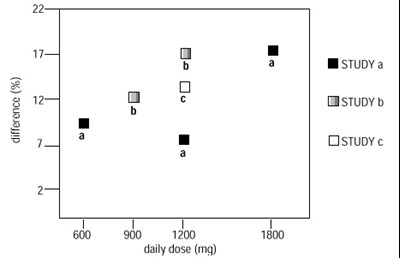
figure 1 - gaba 03

The text may represent the results of a pain management study where the mean pain score is being measured for a group of patients over a period of six weeks. There seems to be a comparison between the effects of a placebo and a medication called Gaboparein taken at a dose of 3600 mg per day. However, the text is incomplete and lacks context, so further interpretation is not possible.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.