Product Images Dofetilide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Dofetilide NDC 69452-131 by Bionpharma Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
graph00005
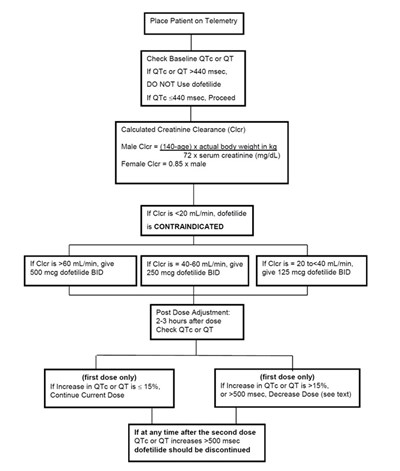
The text provides instructions related to administering dofetilide, a medication used for heart arrhythmias. It outlines the importance of placing the patient on telemetry and checking their baseline QTc or QT. However, it warns against using dofetiide for a patient with a QTc <440 msec. The text offers guidelines for dosing based on the patient's Calculated Creatinine Clearance (Cler) and cautions against giving dofetiide if the Cler is <20 mI/min. Post-Dose adjustment guidelines are provided based on the percentage of increase in QTc or QT after the second dose. The text instructs to discontinue dofetilide if at any point after the second dose QTc or QT increases >500 msec.*
graph0004

This appears to be a graph or chart showing the probability of remaining in normal sinus rhythm (NSR) over time for different dosages of a medication called Dofet. The dosage levels are listed as 125 mcg, 250 mcg, and 500 mcg, taken twice a day (BID). There is also a line indicating the p-values obtained from a Log Rank Test. However, without additional context, it is difficult to determine the specific purpose or implications of this information.*
Figure 1 - graph001
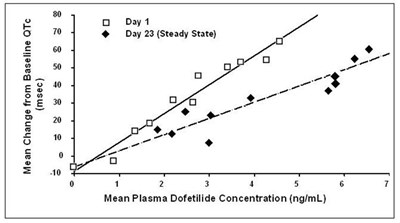
The text describes the mean change in QTc from baseline, measured in millisecond (msec), and the mean plasma concentration of Dofetilide (measured in ng/mL) on Day 1 and Day 23 (Steady State) during a study. No further information is available.*
Figure 2 - graph002
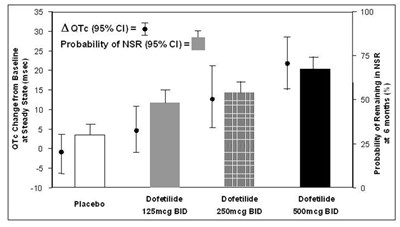
This appears to be a table displaying QTc (a measurement of heart rate) changes from baseline at steady state for different doses of dofetilide medication and a placebo. The table also includes the probability of remaining in normal sinus rhythm (NSR) at 6 months for each dose. There is no information available about the study's purpose, sample size, or other research data.*
125 mcg (0.125 mg) 60's Label - image0001v1

The text cannot be evaluated as it consists of random and unrelated characters and texts.*
250 mcg (0.25 mg) 60's Label - image0002v1

BronPHARMA dispenses 60 capsules of Dofetilide, each containing 250 mcg (0.25 mg) as a treatment for abnormal heart rhythms. The NDC code is 69452-132.17.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


