Product Images Terazosin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Terazosin NDC 69452-333 by Bionpharma Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image description - figure 01
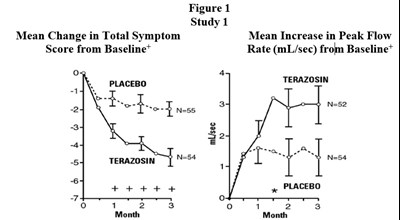
This text provides information on the mean change in total symptom score and the mean increase in peak flow rate (mL/sec) from baseline between the placebo and terazosin groups in a study. The study includes 54 participants and is represented graphically in Figure 1. Additionally, the data is presented over a 3-month period.*
image description - figure 02
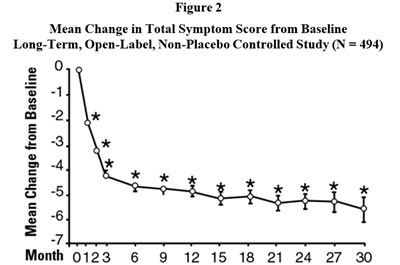
This is a figure showing the mean change in total symptom score from baseline in a long-term, open-label study with 494 participants. The data is presented for different time points up to 30 months.*
image description - figure 03
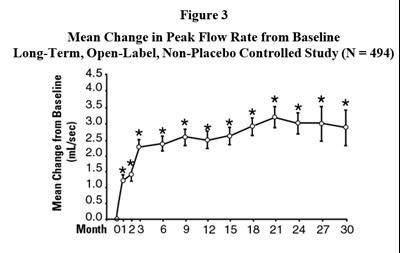
This information shows the mean change in peak flow rate from the baseline in a Long-Term, Open-Label, Non-Placebo Controlled Study involving 494 participants. The data is presented in a graphical format depicting the change in milliliters per second over a period of 30 months.*
image description - image001

This is a description of Terazosin capsules, a prescription medication containing terazosin hydrochloride, USP equivalent to 1mg of terazosin. The usual dosage information is included in the package insert. The capsules should be stored in a tight, light-resistant container at temperatures between 20°C to 25°C (68°F to 77°F), with excursions permitted to 15°C to 30°C (59°F to 86°F). It is essential to protect the capsules from light and moisture. These capsules are distributed by Bionpharma Inc. in Princeton, NJ, and are manufactured in India. Additional codes and identification numbers are listed for tracking purposes.*
image description - image002

This text provides information about Terazosin capsules, USP with each capsule containing Terazosin hydrochloride equivalent to 2 mg of Terazosin. It includes instructions for usual dosage, storage conditions, and the manufacturer details. The capsules should be dispensed in a tight, light-resistant container and protected from light and moisture. The product is distributed by Bionpharma Inc. in Princeton, NJ, and the manufacturing is done in India.*
image description - image003

This is a description of Terazosin capsules, each containing 5 mg of Terazosin hydrochloride, USP. The usual dosage instructions are provided in the package insert. The capsules should be stored in a tight, light-resistant container at a temperature between 20°C to 25°C (68°F to 77°F), with permitted excursions to 16°C to 30°C (59°F to 86°F). It is essential to protect the capsules from light and moisture. The product is distributed by Bionpharma Inc. and manufactured in India.*
image description - image004

This text is a detailed description of Terazosin capsules, a prescription medication containing 10mg of Terazosin hydrochloride. It includes storage instructions, prescribing information, and details of the manufacturer, Bionpharma Inc. The medication is distributed in tight, light-resistant containers to protect it from light and moisture. The text also provides the NDC, Rx only label, dosage information, and the capsule count in the package.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
