Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 45 images provide visual information about the product associated with Pregabalin NDC 69539-013 by Msn Laboratories Private Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
equation - pregabain gault equation
This is a formula used to calculate the Creatinine Clearance Rate (CLCr) of a patient. The formula takes into account the patient's age, weight, and serum creatinine level. The value obtained represents an estimate of the patient's glomerular filtration rate (GFR), which is a measure of how well the kidneys are functioning. The formula includes a correction factor for female patients.*
100mg-1000s - pregabalin 100 mg 1000s count

This is a prescription drug containing 100mg of pregabalin per capsule. It is manufactured by MSN Laboratories in Mahabubnagar, India and distributed by MSN Pharmaceuticals Inc. The usual dosage and prescribing information should be consulted. It is recommended to store the medication in tight, child-resistant containers at a temperature of 15-30°C (59-86°F). The batch number is not provided, but the expiration date is indicated as "12/2016".*
100mg-10s - pregabalin 100 mg 10s carton

This text provides information on Pregabalin Capsules, which is distributed by MSN Pharmaceuticals Inc. The medication guide should be dispensed to each patient for institutional use only. The usual dosage is provided in the accompanying Prescribing Information. Each capsule contains 100mg of pregabalin. The manufacturing company is MSN Laboratories Private Limited, located in Telangana, India. The text also includes the product's NDC number and a lot number.*
100mg-90s - pregabalin 100 mg 90s count

This text contains information about the storage, temperature, dosage, and manufacturer of Pregabalin Capsules containing 100mg of the drug. The manufacturer is MSN Laboratories Private Limited based in Telangana, India and is distributed by MSN Pharmaceuticals Inc. Patients should be dispensed with tight and child-resistant containers along with the accompanying prescribing information and medication guide. The text also contains batch and expiry information.*
150mg-carton - pregabalin 150 mg 10s carton

This is the description of a Pregabalin Capsule medication with packaging details. The medication guide should be given to each patient, and it is for institutional use only. The package contains 100 unit-dose capsules, and they should be stored at a controlled room temperature of 25°C (77°F) with excursions permitted to 15-30°C (59-86°F). Each capsule contains 150 mg of pregabalin. The manufacturer is MSN Laboratories Private Limited located in Telangana, India, and it is distributed by MSN Pharmaceuticals Inc. in Edison, NJ. The package was issued in December 2016.*
150mg-500s - pregabalin 150 mg 500s count

This is a medication containing 150mg of pregabalin. It is manufactured by MSN Laboratories Private Limited, based in Mahabubnagar, Telangana, India. The medication should be stored in a tight, child-resistant container at 25°C (77°F), with permitted excursions to 15-30°C (59-86°F), according to USP Controlled Room Temperature guidelines. It is distributed in the US by MSN Pharmaceuticals Inc. An accompanying prescribing information document and medication guide should be dispensed with the medication. The NDC number is 69539-015-05 and is available as a pack of 500 capsules.*
150mg-90s - pregabalin 150 mg 90s count

This is a medication containing 150mg of pregabalin per capsule. It should be stored at a temperature of 25°C (77°F) with permitted excursions to 15-30°C (59-86°F). The medication should be dispensed in a tight, child-resistant container. The usual dosage is to be determined by accompanying prescribing information. The medication guide should be given to each patient. The batch and expiry date are not available. It is manufactured by MSN Laboratories Private Limited in Telangana, India and distributed by MSN Pharmaceuticals Inc. in Edison, NJ. This product is only available by prescription.*
200mg-1000s - pregabalin 200 mg 1000s count

Store the Progabalin Capsules at a temperature between 15-30°C (59-86°F) as permitted, with excursions to 25°C (77°F). The drug should be dispensed in tight, child-resistant containers. The usual dosage and prescription information can be found in the accompanying medication guide. The product is distributed by MSN Pharmaceuticals Inc. in a batch of 1000 capsules. The manufacturer is MSN Laboratories Private Limited from Mahabubnagar, Telangana - 509216, INDIA. The expiration date for this batch is not available.*
200mg-carton - pregabalin 200 mg 10s carton

This is a description for a medication named Pregabalin Capsules, with a concentration of 200mg per capsule. The package comes with 100 (10 X 10) unit-dose capsules, and a Medication Guide that should be dispensed to each patient. It's for institutional use only, and contains Rx Only prescriptions. The capsules should be stored at 25°C (77°F), with excursions permitted to 15-30°C (59-86°F), as per USP Controlled Room Temperature guidelines. The description suggests that there's accompanying prescribing information, but that's not available in the provided text. The medication is manufactured by MSN Laboratories Private Limited, located in Mahabubnagar (Dt), Telangana in India, while distribution is done by MSN Pharmaceuticals Inc., based in Edison, NJ 08837. The text contains irrelevant characters that don't make sense in this context.*
200mg-90s - pregabalin 200 mg 90s count

This is a description of Pregabalin Capsules, which come in a bottle of 90 capsules. Each capsule contains 200mg of pregabalin. The medication should be stored at 25°C (77°F) with permitted excursions to 15-30°C (59-86°F). It is dispensed in tight (USP), child-resistant containers along with an accompanying medication guide. The usual dosage information is provided in the accompanying prescribing information. The manufacturer is MSN Laboratories Private Limited, based in India, and the medication is distributed by MSN Pharmaceuticals Inc. based in Edison, NJ. Other information provided includes a lot number, expiration date, and M.L.No. 5/MN/TS/2014/FIG issued on 12/2016.*
225mg-1000s - pregabalin 225 mg 1000s count

This is a medication information for Pregabalin capsules containing 225 mg of the drug in each capsule. It is manufactured by MSN Laboratories Private Limited in Mahabubnagar, India and distributed by MSN Pharmaceuticals Inc. in Edison, NJ. The capsules should be stored in tightly closed, child-resistant containers at temperature between 15-30°C (59-86°F) and protected from excursions. Its usual dosage is subject to a doctor's prescription. The medication guide accompanies each patient's prescription. The seems to have jumbled some text in the description.*
225mg-10s - pregabalin 225 mg 10s carton

The text describes the medication Pregabalin Capsules, with a dosage of 225mg per capsule. The medication is for institutional use only, with accompanying medication guides to be given to each patient. It also gives the NDC code as 69539-017-06 and lists the manufacturer and distributor information. The capsules come in a 100 (10 X 10) unit-dose capsule package and should be stored at 25°C with excursions permitted to 15-30°C. The usual dosage information can be found in the accompanying Prescribing Information.*
225mg-90s - pregabalin 225 mg 90s count

This is a label for pregabalin capsules. It provides information on the storage of the medication and the usual dosage. The medication is dispensed in child-resistant containers and comes with an accompanying medication guide. The manufacturers are MSD Laboratories Private Limited in India and it is distributed by MSN Pharmaceuticals Inc. in the US. The label also includes batch and expiration information.*
25mg-1000s - pregabalin 25 mg 1000s count

This is a prescription medication called Pregabalin Capsules, containing 25mg each. Patients should store the capsules in a dry place, at a temperature of 25°C or 77°F, with fluctuations permitted between 15-30°C or 59-86°F. Tight child-resistant containers should be used to dispense this medication. The accompanying prescribing information and medication guide should be given to each patient. This medication is sold in containers containing 1000 capsules. It is manufactured by MSN Laboratories Private Limited, based in India, and distributed by MSN Pharmaceuticals Inc. based in Edison, NJ, USA. No batch or expiry dates are provided.*
25mg-10s - pregabalin 25 mg 10s carton

This is a description of Pregabalin Capsules medication. Each capsule contains 25mg of pregabalin. The accompanying medication guide must be dispensed to each patient. It is for institutional use only. The usual dosage is outlined in the accompanying prescription information. Each package contains 100 (10 x 10) unit-dose capsules. They should be stored at 25°C (77°F) and may be allowed excursion of 15-30°C (59-86°F) according to the USP Controlled Room Temperature. They were manufactured in Mahabubnagar (Dt), Telangana, India, by MSN Laboratories Private Limited and distributed by MSN Pharmaceuticals Inc. in Edison, NJ 08837. This information was issued in December 2016.*
25mg-90s - pregabalin 25 mg 90s count

Store the Pregabalin capsules at room temperature of 25°C (77°F) with permissible temperature excursions between 15-30°C (59-86°F). The capsules should be dispensed in tight, child-resistant containers as per USP. The quantity of pregabalin in each capsule is 25 mg. Patients should refer to the attached prescribing information for the usual dosage instructions. The capsules come in a pack of 90 and should be distributed along with the medication guide. The batch number and expiration date are not available. The capsules are manufactured by MSN Laboratories Private Limited in India and distributed by MSN Pharmaceuticals Inc. in Edison, NJ. It is only available by prescription.*
300mg-10s - pregabalin 300 mg 10s carton

This is a description of a medicine called Pregabalin Capsules that come in a pack of 100 unit-dose capsules. It contains 300 mg of pregabalin and is meant to be dispensed with a medication guide to each patient. The medication guide should be read alongside the accompanying Prescribing information. It is manufactured by MSN Laboratories Private Limited located in Mahabubnagar (Dt), Telangana, India and distributed by MSN Pharmaceuticals Inc. located in Edison, NJ 08837. It is for institutional use only and should be stored at 15-30°C (59-86°F).*
300mg-90s - pregabalin 300 mg 90s count

This is a description of Pregabalin capsules. The capsules contain 300mg of pregabalin and are manufactured by MSN Laboratcne{ Private Limited in India. They should be stored at 25°C (77°F) with excursions permitted to 1(5-30“)(: (59-86°F) and dispensed in tight, child-resistant containers. The product should be regulated under USP Controlled Room Temperature. Each purchase of the 90 capsules should be accompanied by a Medication Guide to provide proper use guidance to patients. Batch numbers are provided, but the Expiry date is not available in the text.*
50mg-1000s - pregabalin 50 mg 1000s count

This is a prescription medication guide for Pregabalin Capsules containing 50mg of the active ingredient per capsule, manufactured by MSN Laboratories Private Limited. The capsules are dispensed in child-resistant containers and should be stored at a temperature between 15-30°C (59-86°F) according to USP Controlled Room Temperature guidelines. The medication is distributed by MSN Pharmaceuticals Inc. and comes with an accompanying prescribing information and medication guide. The batch must be dispensed with the expiry of 1000 capsules marked as Rx Only.*
50mg-10s - pregabalin 50 mg 10s carton

Pregabalin Capsules is a medication that comes in a pack of 100 (10 x 10) unit-dose capsules, with a usual dosage of 50 mg per capsule. It's for institutional use only and should be dispensed to the patient with the accompanying medication guide. The medicine should be stored at 25°C (77°F) with limited excursions to 15-30°C (59-86°F). Pregabalin Capsules are distributed by MSN Pharmaceuticals Inc. and manufactured by MSN Laboratories Private Limited, located in Mahabubnagar (Dt), Telangana, India.*
50mg-90s - pregabalin 50 mg 90s count

Store the Pregabalin Capsules at a temperature of 25°C (77°F) with permitted excursions within 15-30°C (59-86°F), as per USP Controlled Room Temperature guidelines. Dispense the medication in tight (USP) and child-resistant containers. Each capsule contains 50mg of pregabalin. Quantity (MLNo.) is 5IMNITS/2014/FIG, and it was issued in 12/2016. The medication guide should be dispensed to the patient along with 90 capsules. The medicine is manufactured by MSN Laboratories Private Limited and distributed by MSN Pharmaceuticals Inc. The batch number and expiry date are not available.*
75mg-1000s - pregabalin 75 mg 1000s count

This text provides information about the storage, manufacturing, and distribution of 75mg Pregabalin Capsules by MSN Laboratories Private Limited. It includes guidelines for storing the medication, dosage instructions, manufacturing details, batch number, and expiry date. The medication should be dispensed in child-resistant containers with a prescription, and each patient should receive an accompanying medication guide.*
75mg-10s - pregabalin 75 mg 10s carton

Pregabalin Capsules is a prescription drug that comes with a Medication Guide. It contains 75 mg of pregabalin and comes in a pack of 100 (10 X 10) Unit-dose Capsules. The drug is only for institutional use and must be stored at 25°C (77°F). The usual dosage is provided in the Prescribing Information. The manufacturer is MSN Laboratories Private Limited based in Mahabubnagar (Dt), Telangana - 509216, INDIA, and is distributed by MSN Pharmaceuticals Inc. based in Edison, NJ 08837. The package was issued in December 2016.*
75mg-90s - pregabalin 75 mg 90s count

This is a label for pregabalin capsules containing 75mg each. The capsules should be dispensed in child-resistant containers and come with an accompanying medication guide. The capsules should be stored at 25°C with excursions permitted to 15-30°C. The label indicates the manufacturer, batch number, expiration date, and distribution information.*
fig-01 - pregabalin figure 01

This is a chart that shows the percentage of patients who improved with different doses of Pregabalin and placebo. It also shows the percentage of improvement in pain from baseline.*
fig-02 - pregabalin figure 02

This is a graph plotting the percentage of patients who improved on Pregabalin 100mg taken three times a day versus Placebo, where the axis represents improvement in pain from baseline. The graph shows that Pregabalin was more effective in improving pain compared to Placebo.*
fig-03 - pregabalin figure 03
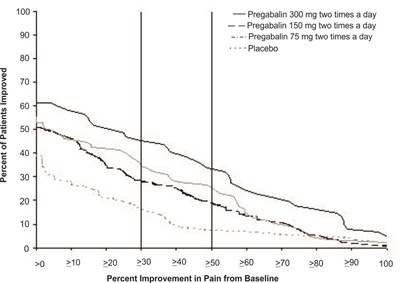
This is a chart that shows the percentage of patients improved based on different doses of Pregabalin compared to placebo. It also shows the percentage improvement in pain from baseline.*
fig-04 - pregabalin figure 04

The text represents the results of a study or experiment for a medication called pregabalin in improving pain. The graph shows the percentage of patients who improved in pain after taking pregabalin 200mg or 100mg three times a day compared to a placebo. The x-axis shows the percentage of improvement in pain from baseline, while the y-axis represents the percentage of patients who improved.*
fig-05 - pregabalin figure 05

This appears to be a chart showing the percent of patients who improved with pregabalin at different dosages compared to a placebo, measured by the percent improvement in pain from baseline. The dosages shown are 100mg three times a day and 50mg three times a day.*
fig-06 - pregabalin figure 06

This appears to be a table showing responder rates (%) for different dosages and frequencies of medication (likely for a study). The table includes percentages for each category (ranging from 10% to 60%) and some information on the study (e.g., Study E1, Study E3). There are also dosage amounts (50mgday, 150mgday, etc.) and frequency information (BID, TID). The asterisk denotes statistically significant results versus placebo. However, further information or context is necessary to ascertain the nature and purpose of the study or medication being investigated.*
fig-09 - pregabalin figure 09

This is a graph that shows the percentage of patients who improved based on different daily doses of pregabalin and a placebo. The x-axis shows the percentage of improvement in pain from baseline while the y-axis shows the percentage of patients who had this level of improvement. However, there is no clear indication of what condition this improvement was being evaluated for.*
fig-11 - pregabalin figure 11

This document appears to be a graph displaying the percentage of subjects who improved from baseline to week 12 using pregabalin versus placebo. The percentage of improved subjects ranges from 0 to 100, and the graph displays improvement intervals of 10.*
fig-12 - pregabalin figure 12

This table shows the percentage of subjects who showed improvement during a study that lasted for 16 weeks. The study compared the effects of Pregabalin versus a Placebo. The table shows percentages in intervals of 10, from 0 to 80. It also includes a single data point above 80. There are no further details provided to explain the nature and purpose of the study or what the terms "BOCF B" mean.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.











