Product Images Sodium Phenylacetate And Sodium Benzoate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Sodium Phenylacetate And Sodium Benzoate NDC 69784-810 by Woodward Pharma Services Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Chemical Structure - ammonul 01

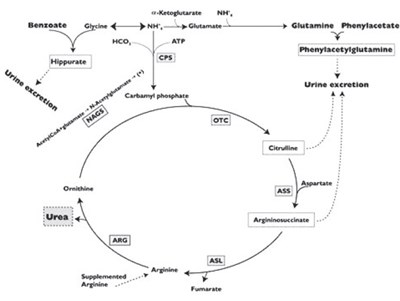
The text appears to be chemically related and contains some misspellings. It is not available to provide a useful description without context or clarification.*
PRINCIPAL DISPLAY PANEL - 50 mL Vial Carton - ammonul 04

Sodium Phenylacetate and Sodium Benzoate Injection is a sterile, non-pyrogenic, single-dose vial that is only used for IV administration. It contains 100 mg of sodium phenylacetate and 100 mg of sodium benzoate in 1 mL of water for injection, and requires dilution with sterile dextrose injection before administration. This medication must be administered through a central line, as administration through a peripheral line may cause burns. It is manufactured by Woodward Pharma and stored at temperatures permitted between 15°C - 30°C (59°F - 86°F). The accompanying package insert should be consulted for dosage information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.