Product Images Dexamethasone Sodium Phosphate
View Photos of Packaging, Labels & Appearance
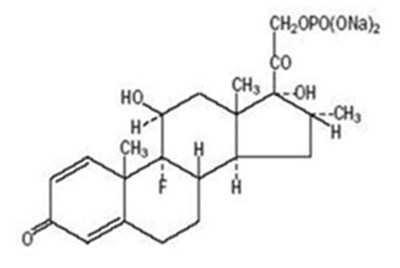
- Molecular structure - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 01
- 1 mL Container label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 02
- 1 mL Carton label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 03
- 5 mL Container label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 04
- 5 mL Carton label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 05
- 30 mL Container label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 06
- 30 mL Carton label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 07
Product Label Images
The following 7 images provide visual information about the product associated with Dexamethasone Sodium Phosphate NDC 70069-022 by Somerset Therapeutics, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1 mL Container label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 02

This is a description of Dexamethasone Sodium Phosphate Injection, USP. It is indicated for intravenous or intramuscular use and comes in a single dose vial of 1mL. The medication is sensitive to heat and should not be autoclaved, and it requires protection from light. It should be stored at 20° to 25°C (68° to 77°F), and the package insert should be consulted for additional routes. The medication is manufactured by Wintac Limited in Bangalore, India, and is distributed by Somerset Therapeutics LLC in Somerset, NJ.*
1 mL Carton label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 03

Dexamethasone Sodium Phosphate Injection, USP, is a medication used for intravenous or intramuscular administration. Each 1 mL of the injection contains Deamethasone sodium phosphate (437 mg equivalent to 4 mg), Sodium Sulfite 1 mg, Berayl Acorol 10 mg, Water for Injection, Sodium Citrate for tonicity, pH adjusted with Glacial Acetic Acid or Sodium Hydroxide. The medication should not be autoclaved. The package insert should be consulted for directions of use. It can be stored at 20 to 25°C (68 to 77°F) and is available in 251 mL single dose vials. The text also includes the product's NDC number, which is 70069-022-25.*
5 mL Container label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 04

Dexamethasone Sodium Phosphate Injection is a prescription-only medicine that is available in a 5mL multiple dose vial with a strength of 20mg/5mL. The medication is meant for intravenous or intramuscular use and should be protected from light and not autoclaved. It is manufactured by Wintac Limited in Bangalore, India, and is sold by Somerset Therapeutics LLC based in Somerset, NJ. The drug is sensitive to heat and should be stored at 20° to 25°C. Please refer to the package insert for additional routes for administration.*
5 mL Carton label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 05

This appears to be a product description for Dexamethasone Sodium Phosphate Injection, USP, with each mL containing 437 mg equivalent to 0.4g of the active ingredient. It also lists other ingredients and storage instructions. It is unclear what the numbers and letters at the beginning and end are referring to.*
30 mL Container label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 06

Dexamethasone Sodium Phosphate Injection is a medication given intravenously, intramuscularly, intra-articularly, soft tissue or intralesionally, with 120mg/30mL, which essentially stands for 4mg/r. It comes in a 30 mL multiple dose vial which contains Dexamethasone sodium phosphate, Sodium Sulphate, Benzyl Alcohol, Citric acid, and Sodium Hydroxide. This medication should not be autoclaved and should be protected from light. It should be stored at an average temperature of 20°to 25°C (68° to 77°F). The product is manufactured by Wintac Limited in Bangalore, India and imported into the United States by Therspeutia Inc.*
30 mL Carton label - 91a391c4 3cb2 4161 aeb7 aec21fab2b46 07

This is a description of a pharmaceutical product. The product is an injection called Dexamethasone Sodium Phosphate USP, which is used for intravenous, intramuscular, intra-articular, soft tissue or inhalational purposes. The injection comes in a 25430 Multiple Dose container and should be protected from light. It is sensitive to heat, so it should not be autoclaved. The package insert should be consulted for proper usage directions. Storage should be at 20° to 25°C (68°-77°F) according to USP Controlled Room Temperature regulations.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.