Product Images Cisatracurium Besylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Cisatracurium Besylate NDC 70069-141 by Somerset Therapeutics, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
d1a742e1 8c8c 4de0 9ea0 9c38961d18b6 02
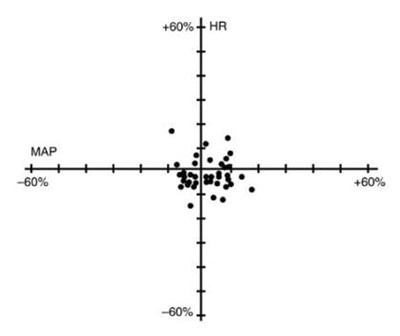
This seems to be a visual representation of some numerical data indicating percentage changes. The text shows the labels "MAP" and "HR" indicating that there may be two different metrics being compared. The percentage values indicate changes which can be positive, negative or as high as 460% which may indicate a significant increase. However, without further context, it is difficult to interpret what the data represents.*
d1a742e1 8c8c 4de0 9ea0 9c38961d18b6 04

Cisatracurium Besylate Injection is a sterile and nonpyrogenic paralyzing agent used for intravenous injection. The package includes a 5 mL single-dose vial that contains 10 mg/5 mL (2 mg/mL) of the injection. As it can cause respiratory arrest, facilities for artificial respiration should always be available. It should be stored between 2°C to 8°C (36°F to 46°F) and protected from light. Use within 21 days of removal from the refrigerator, even if reused the refrigerator. Any package which has passed its expiration date should be discarded. This drug is manufactured by Somerset Therapeutics, LLC in India.*
d1a742e1 8c8c 4de0 9ea0 9c38961d18b6 06
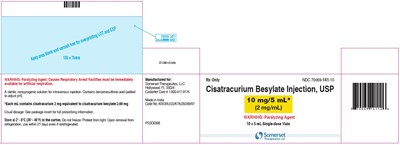
This is a warning label for a drug called Cisatracurium Besylate Injection, which is a paralyzing agent that can cause respiratory arrest. It is only available under prescription and manufactured by Somerset Therapeutics LLC. The usual dosage is mentioned, and it should be stored at a specific temperature range and protected from light. A package insert contains prescribing information. There are 10 single-dose vials each with 5mL of medication available under NDC 70069-141-10. The text is partially readable, but there are misplaced characters and non-English characters, which may make it difficult to understand the complete details.*
d1a742e1 8c8c 4de0 9ea0 9c38961d18b6 07

This is a warning label for the drug Cisatracurium Besylate injection, USP. It advises facilities to have immediate access to artificial respiration as the medication may cause respiratory issues. The drug should be stored between 2°C to 8°C (36 to 46°F) and should not be frozen or exposed to light. It also contains information about the size of the vial which is 13.5 x 30mm.*
d1a742e1 8c8c 4de0 9ea0 9c38961d18b6 08
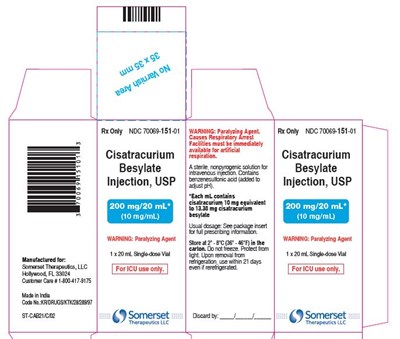
This is a warning label for an injection of Cisatracurium Besylate, which is a paralyzing agent used during medical procedures. The label warns that the drug can cause respiratory arrest and that facilities for artificial respiration must be immediately available. The usual dosage is not provided and the label states that it is for single use only. The manufacturer's contact information is also provided.*
d1a742e1 8c8c 4de0 9ea0 9c38961d18b6 09

Cisatracurium Besylate Injection is a non-pyrogenic injection used intravenously. However, it is a paralyzing agent that can lead to respiratory arrest. It is advised to have facilities available for artificial respiration. Each mL of the injection has cisatracurium equivalent to 13.38 mg cisatracurium besylate. The dosage should be taken as specified in the prescription. The injection should be stored at 20-25°C or 68-77°F and protected from light. The manufacturer of the injection is Somerset Therapeutics, LLC based in Florida. The text also includes product codes and warnings.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


