Product Images Bupivacaine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Bupivacaine Hydrochloride NDC 70069-752 by Somerset Therapeutics, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - 86b0d5d5 6a92 419f 9060 25598fe19aae 02

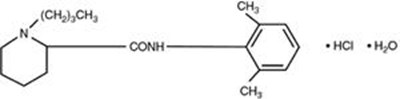
This is a description of a medication called Bupivacaine Hydrochloride Injection, USP 0.25%. It is used for infiltration and nerve block procedures. This medication should not be used for caudal, epidural, or intrathecal anesthesia. It comes in a 50 mL multiple-dose vial, and each mL contains 2.5 mg of bupivacaine HCI. The solution is made isotonic with NaCl in water for injection, and the pH is adjusted with NaOH or HCI. The dosage information can be found in the package insert. It should be stored at 20° to 25°C (68° to 77°F). The manufacturer of this medication is Somerset Therapeutics, LLC, based in Hollywood, FL 33024. It is made in India. The neutral code number is TN/DRUGS/616/1996. There is also a lot number and an expiration date, but they are not readable in the provided text. The dimensions of the area for overprinting are 325x15mm, and it should be kept blank and varnish free.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.