Product Images Fulvestrant
View Photos of Packaging, Labels & Appearance
- 13 - fulvestrant injection 1
- 10 - fulvestrant injection 10
- 1 - fulvestrant injection 11
- fig 11 - fulvestrant injection 12
- fig 12 - fulvestrant injection 13
- fig 13 - fulvestrant injection 14
- 1 - fulvestrant injection 15
- 1 - fulvestrant injection 16
- 1 - fulvestrant injection 17
- 14 - fulvestrant injection 2
- 15 - fulvestrant injection 3
- 15 - fulvestrant injection 4
- 17 - fulvestrant injection 5
- 16 - fulvestrant injection 6
- 66 - fulvestrant injection 7
- 10 - fulvestrant injection 8
- FIG 8 - fulvestrant injection 9
Product Label Images
The following 17 images provide visual information about the product associated with Fulvestrant NDC 70121-1463 by Amneal Pharmaceuticals Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 - fulvestrant injection 10
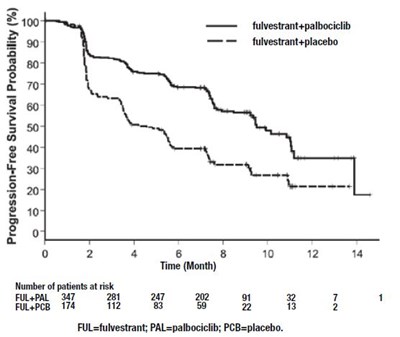
The text seems to be a comparison of two treatments for cancer, fulvestrant alone or fulvestrant in combination with palbociclib. The graph shows the probability of progression-free survival (%) over the course of several months for each treatment group. The numbers below the graph suggest the number of patients at risk over time for each treatment group.*
1 - fulvestrant injection 11
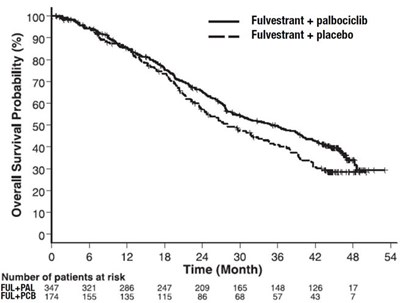
This document reports survival probabilities of two treatment groups (Fulvestrant + Palbociclib and Fulvestrant + Placebo) for a specific period of time (0 to 54 months) based on the number of patients at risk. The percentage values of survival probabilities are not available due to the 's inability to read information.*
fig 12 - fulvestrant injection 13

This is a survival plot of two treatment groups, Fulvestrant plus Abemaciclib (N=446) and Fulvestrant plus Placebo (N=223), showing the percentage of survival probability over time (in months) on the x-axis. Unfortunately, additional information is not available due to errors in the text.*
1 - fulvestrant injection 16

Fulvestrant Injection is a medication that comes in a 250 mg/5 mL (50 mg/mL) dosage and is only for intramuscular use. It contains two single-dose prefilled syringes, and both must be used to receive the 500 mg dose. The medication is indicated to treat breast cancer. Some of the important information available include the barcode, expiry date, and other descriptive details.*
1 - fulvestrant injection 17

This is a description for a medication called Fulvestrant Injection. The medication comes in a pre-filled syringe, with each syringe containing 250 mg/5 mL or 50 my/ml of the medication. The syringe is for intramuscular use only and should be protected from light. To receive the full 600 mg dose, both single-dose prefilled syringes should be administered. The manufacturer is Amneal Pharmaceuticals and the medication has an NDC number of 70121-1463-2. The rest of the text appears to be a mix of random characters and not useful in describing the medication.*
66 - fulvestrant injection 7
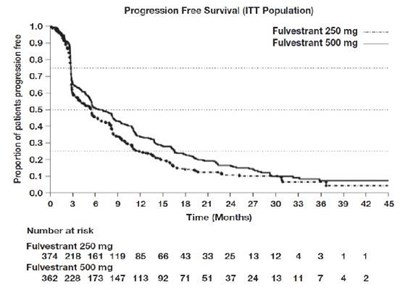
This is a graph that shows the progression-free survival of patients who were given either fulvestrant 250 mg or fulvestrant 500 mg. The graph indicates the number of patients at risk at different time intervals ranging from 0 to 45 months. The x-axis shows the time intervals while the y-axis shows the number of patients at risk.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.










