Product Images Lioresal (baclofen)
View Photos of Packaging, Labels & Appearance
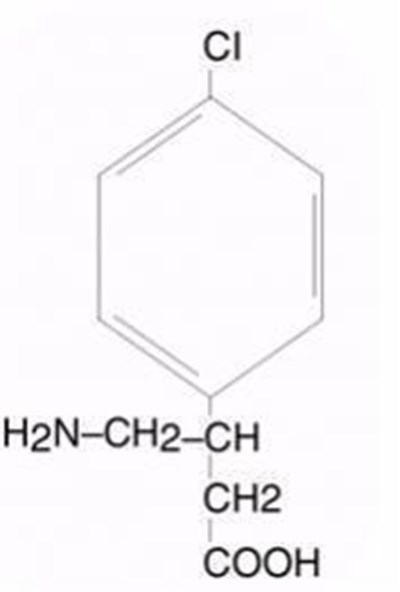
- Chemical Structure - lioresal intrathecal baclofen injection 1
- figure 3 - lioresal intrathecal baclofen injection 10
- figure 4 - lioresal intrathecal baclofen injection 11
- figure 5 - lioresal intrathecal baclofen injection 12
- figure 6 - lioresal intrathecal baclofen injection 13
- drug prep kit two syringes - lioresal intrathecal baclofen injection 14
- drug prep kit one syringe - lioresal intrathecal baclofen injection 15
- 8561 - lioresal intrathecal baclofen injection 16
- 8562 - lioresal intrathecal baclofen injection 17
- 8564 - lioresal intrathecal baclofen injection 18
- 8565 - lioresal intrathecal baclofen injection 19
- Figure 1 - lioresal intrathecal baclofen injection 2
- 8566 - lioresal intrathecal baclofen injection 20
- 856X refill kit - lioresal intrathecal baclofen injection 21
- 8563s ifc - lioresal intrathecal baclofen injection 22
- 50mcg vial label - lioresal intrathecal baclofen injection 23
- 5x20ml ifc - lioresal intrathecal baclofen injection 24
- 2000mcg vial - lioresal intrathecal baclofen injection 25
- 500mcg vial label - lioresal intrathecal baclofen injection 26
- 10xs5ml ifc - lioresal intrathecal baclofen injection 27
- Figure 2 - lioresal intrathecal baclofen injection 3
- Figure 3 - lioresal intrathecal baclofen injection 4
- Figure 4 - lioresal intrathecal baclofen injection 5
- Figure 5 - lioresal intrathecal baclofen injection 6
- open here - lioresal intrathecal baclofen injection 7
- figure 1 - lioresal intrathecal baclofen injection 8
- figure 2 - lioresal intrathecal baclofen injection 9
Product Label Images
The following 27 images provide visual information about the product associated with Lioresal (baclofen) NDC 70121-2503 by Amneal Pharmaceuticals Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
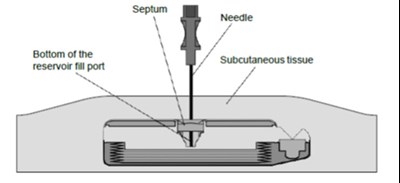
figure 5 - lioresal intrathecal baclofen injection 12

The text appears to be a simple label or title for a reservoir fill port template. No further information or description is available.*
drug prep kit two syringes - lioresal intrathecal baclofen injection 14
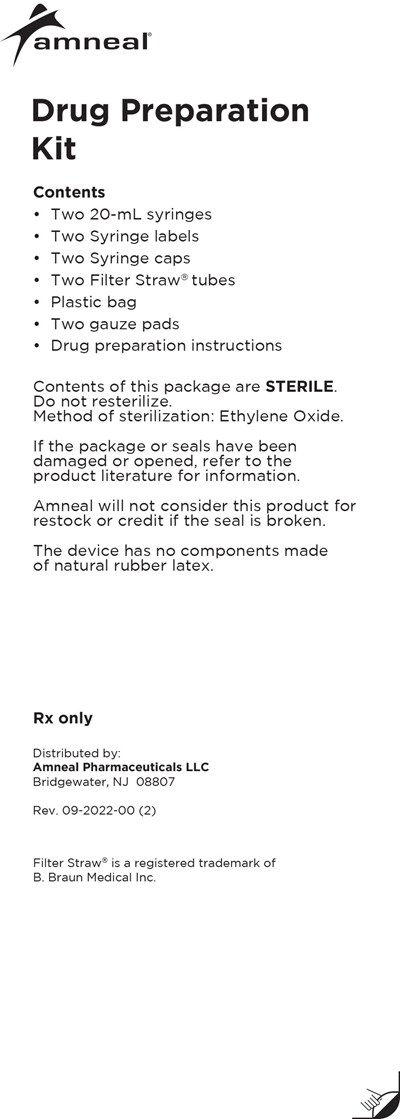
The Amneal® Drug Preparation Kit contains sterilized supplies for preparing drugs for medical use. It includes two 20-mL syringes, syringe labels, syringe caps, and Filter Straw® tubes along with other components. The package is sterile and sterilized via ethylene oxide. The use of the product should only be done by Rx only. Amneal Pharmaceuticals LLC distributes the kit and provides instructions for use.*
drug prep kit one syringe - lioresal intrathecal baclofen injection 15

This is a description of a drug preparation kit manufactured by Amneal. The kit contains a 20-mL syringe, syringe label, syringe cap, Filter Straw® tube, plastic bag, two gauze pads, and drug preparation instructions. The contents are sterile and cannot be resterilized. It is distributed by Amneal Pharmaceuticals LLC in Bridgewater, NJ, and has no components made of natural rubber latex. The kit is Rx only, and Filter Straw® is a registered trademark of B. Braun Medical Inc.*
8561 - lioresal intrathecal baclofen injection 16

This is a description of Lioresal Intrathecal Baclofen Injection refill kit by Amneal Pharmaceuticals. The kit contains a 20-mL syringe, 22-gauge needles, a filter straw tube, a 0.2-micron filter, drug prescribing information, and accessories. It is used with SynchroMed Infusion Systems and does not require refrigeration or heating sterilization. The kit has been sterilized by ethylene oxide gas and should not be used if seals or packages are damaged. The device has no components made of natural rubber latex. Individual kits within this package may have a later use-by date than the one listed above.*
8562 - lioresal intrathecal baclofen injection 17

This is a product description of a refill kit for Lioresal Intrathecal 8562, which is a baclofen injection. The kit is designed for use with SynchroMed Infusion Systems and contains two 5-mL Lioresal Intrathecal ampules, a 20-mL syringe, an extension set with a clamp, two 22-gauge noncoring needles, a template, a Filter Straw tube, a 0.2-micron filter, and drug preparation instructions, as well as refill instructions and drug prescribing information. The kit is sterilized by ethylene oxide gas and does not require refrigeration, but should not be stored above 86°F (30°C) or frozen. The kit does not contain any components made of natural rubber latex.*
8564 - lioresal intrathecal baclofen injection 18

This is a Lioresal Intrathecal Refill Kit used with SynchroMed Infusion Systems for administering baclofen injection directly into the spinal cord. The kit includes 20 mL of 40 mg/20 mL baclofen solution, a drug box consisting of an extension set, 20-mL syringe, two 22-gauge noncoring needles, a template filter straw tube, a 0.2-micron filter, accessories, drug preparation instructions, and prescribing information. The kit has been sterilized by ethylene oxide gas and should be used before a certain date specified on the package. It should not be used if the seals or packages are damaged and should be stored at temperatures below 86 °F (30 °C) and not frozen.*
8565 - lioresal intrathecal baclofen injection 19

Lioresal Intrathecal Refill Kit is used with SynchroMed Infusion Systems. The kit consists of a drug box, drug preparation kit, extension set with a clamp, and accessories. The total volume of the kit is 40 mL divided into two 20-mL ampules. The contents of the kit are sterilized by ethylene oxide gas and do not require refrigeration. The package has a later use-by date than the one listed above. For proper use, read and follow all enclosed instructions.*
8566 - lioresal intrathecal baclofen injection 20

The text seems to be describing a medication called Lioresal Intrathecal which comes in a refill kit for use with a SynchroMed Infusion System. The refill kit contains two 20-mL ampules of baclofen injection solution, a 20-mL syringe, extension set with a clamp, 0.2-micron filter, and other accessories. The package should not be used if damaged and should not be stored above 86 °F (30 °C) or frozen. It also warns that the medication has a later use-by date than the one listed above, and provides contact information for the manufacturer.*
856X refill kit - lioresal intrathecal baclofen injection 21

The Amneal Refill Kit 856X contains various sterile medical supplies for use in medical procedures. These supplies include a syringe, gauze pads, extension set with clamp, alcohol preps, template, exam gloves, a 0.2-micron filter, two noncoring needles, fenestrated drape, and Povidone Iodine swabs. It is important not to resterilize contents and to refer to product literature if the package or seals are damaged or opened. This device has no natural rubber latex components and distributed by Amneal Pharmaceuticals LLC in Bridgewater, NJ. Note that Amneal will not accept restocks or credit if the seal is broken.*
8563s ifc - lioresal intrathecal baclofen injection 22

Lioresal® Intrathecal is a baclofen injection available in a sterile solution for intrathecal injection only. The text also appears to contain random characters and codes that do not provide additional relevant information.*
5x20ml ifc - lioresal intrathecal baclofen injection 24

Lioresal® Intrathecal is a sterile solution for intrathecal injection only, used for the treatment of severe spasticity. The code "9Z1" is printed on the box, and the product contains 10mg/20mL. The other text is unclear and not readable.*
2000mcg vial - lioresal intrathecal baclofen injection 25

Lioresal Intrathecal is a baclofen injection used for intrathecal injection. It is a sterile solution with 10 mg/5 mL (2000 mcg/mL). This medication is distributed by Amneal Pharmaceuticals and is only available with a prescription. The NDC code for this medication is 70121-2494-1. The label also includes information on the product's expiration date.*
10xs5ml ifc - lioresal intrathecal baclofen injection 27

This is a label for a medication called Lioresal Intrathecal, which is a sterile solution for intrathecal injection containing ten 5 mL ampules. It is distributed by Amneal Pharmaceuticals. The label contains a GTIN, SERIAL, LOT, and EXPIRY number, and is for Rx use only. Other than that, the text is not comprehensible as it contains some invalid characters and symbols.*
figure 2 - lioresal intrathecal baclofen injection 9

This text provides instructions on aligning the refill template with the SynchroMed Il pump by matching their right edges. It is likely part of a larger set of directions or user manual for the device.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.