Product Images Clopidogrel Bisulfate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Clopidogrel Bisulfate NDC 70400-101 by Sterisyn Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure1 - clopidogrel fig
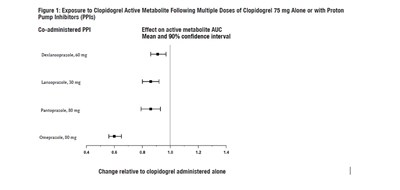
This is a figure that shows how the exposure to the active metabolite of Clopidogrel changes when it is taken alone or in combination with different proton pump inhibitors (PPIs), namely Deslansoprazole, Lansoprszole, Pantoprazole and Omeprazole. The figure presents the effect of each PPI on the active metabolite's AUC, represented by the mean and 90% confidence interval. It also shows the change in relation to the active metabolite of Clopidogrel alone.*
figure3 - clopidogrel fig2
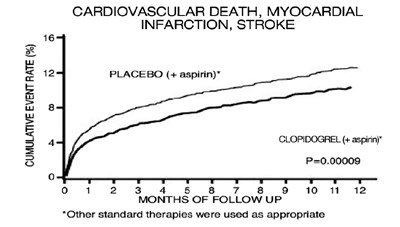
This appears to be a visual representation of a clinical trial comparing the effectiveness of clopidogrel and aspirin versus a placebo plus aspirin in preventing cardiovascular death, myocardial infarction, and stroke. The cumulative event rate is shown over the course of 12 months of follow-up. Standard therapies were also used as appropriate.*
figure4 - clopidogrel fig4
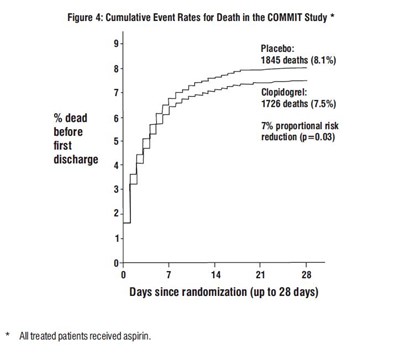
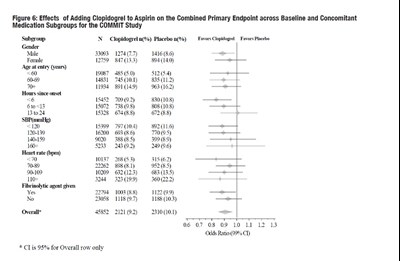
The text is describing the results of the COMMIT study, where patients received either placebo or clopidogrel, in addition to aspirin. It shows the cumulative event rates for death before discharge, with the placebo group having 8.1% deaths and the clopidogrel group having 7.5% deaths. The study found a 7% proportional risk reduction with clopidogrel, with p-value 0.03. The data is also presented in a figure with days since randomization on the x-axis and the percentage of dead patients on the y-axis.*
figure5 - clopidogrel fig5
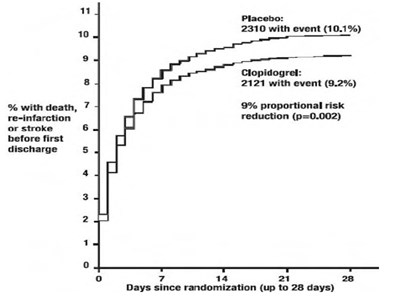
This text appears to be a statistical analysis of two treatments - Placebo and Clopidogrel - used in a clinical trial. The text includes the number of events (deaths or incidences of re-infarction or stroke) that occurred with each treatment, and the proportional risk reduction associated with using Clopidogrel versus Placebo. The data also includes the percentage of patients with death, the reduction in this percentage, and the p-value associated with this reduction. Finally, the data includes the number of days since randomization (up to 28 days).*
figure7 - clopidogrel fig7
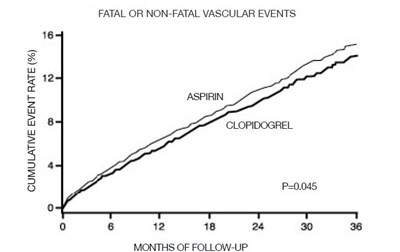
This appears to be a chart or table comparing the cumulative event rate of fatal or non-fatal vascular events for two types of medication, aspirin and clopidogrel, at different time intervals (6, 12, 18 and 24 months). The number of participants (n) and symbols (@ and &) are also included. No further information is available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.