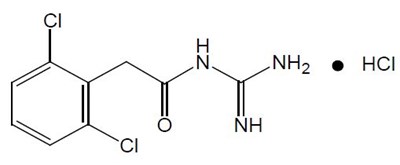
Product Images Guanfacine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Guanfacine NDC 70436-039 by Slate Run Pharmaceuticals, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1mg-100ct.jpg - 1mg 100ct

This is a medication, Guanfacine Extended-Release Tablets, manufactured by Yichang Humanwell Oral Solid Dosage Plant in China and distributed by Slate Run Pharmaceuticals in Columbus, Ohio. Each tablet contains 1 mg of guanfacine hydrochloride, USP, and should not be crushed, chewed, or broken before swallowing. The package insert contains complete prescribing information, and the tablets must be stored at 20-25°C (68-77°F) and dispensed in a tight, light-resistant container with a child-resistant closure. The text at the end appears to be a random assortment of characters and is not informative.*
2mg-100ct - 2mg 100ct

Each tablet of this medicine contains 2mg of guanfacine hydrochloride, USP. It is an extended-release drug and its complete prescribing information can be found in its package insert. The tablets should not be chewed, crushed or broken before swallowing. They should be kept at a temperature range of 20°C to 25°C and dispensed in a tight, light-resistant container out of children's reach. The manufacturer is Yichang Humanwell Oral Solid Dosage Plant, China 443112 and it is distributed by Slate Run Pharmaceuticals, LLC, Columbus, Ohio.*
3mg-100ct - 3mg 100ct

This is a description for Guanfacine Extended-Release Tablets, manufactured by Yichang Humanwell Oral Solid Dosage Plant, Yichang, Hubei, China 443112 and distributed by Slate Run Pharmaceuticals, LLC, Columbus, Ohio 43215. The Tablets come in a secure package containing 100 Tablets, with each extended-release tablet containing 3 mg guanfacine as guanfacine hydrochloride, USP. Usual Dosage: SEE PACKAGE INSERT FOR COMPLETE PRESCRIBING INFORMATION. Tablets should not be crushed, chewed or broken before swallowing. Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required). Keep this and all drugs out of the reach of children.*
4mg-100ct - 4mg 100ct

Each extended-release tablet contains 4 mg of guanfacine hydrochloride, USP. The tablets should be stored at 20° to 25°C (68° to 77°F) and dispensed in a tight, light-resistant container with a child-resistant closure. They should not be crushed, chewed or broken before swallowing. The drug is manufactured by Yichang Humanwell Oral Solid Dosage Plant in China and distributed by Slate Run Pharmaceuticals in Columbus, Ohio.*
figure-1 - figure 1

This appears to be a graph or chart displaying the time (in hours) and dosages (in milligrams) for two types of guanfacine medication: an extended-release form and an immediate-release form. The dosages listed are 1mg QD for both forms. There is no meaningful context or explanation provided beyond this.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.