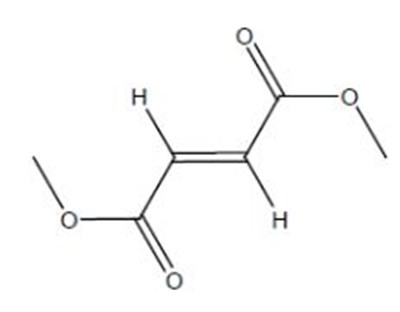
Product Images Dimethyl Fumarate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Dimethyl Fumarate NDC 70512-853 by Sola Pharmaceuticals, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
70512-852-14 120 mg carton - 70512 852 14 120 mg carton
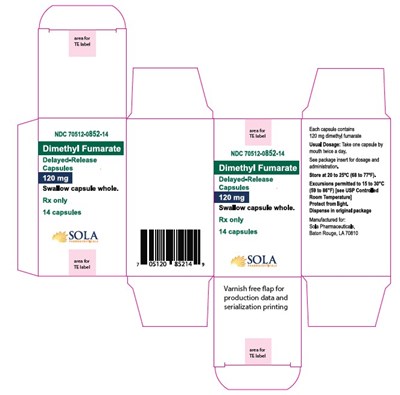
This appears to be a product description for Dimethyl Fumarate capsules which are delayed-release and come in a bottle with 14 capsules. The recommended usage is to swallow the capsule whole. However, due to the poor quality of the text, some parts are not readable and the description may not be accurate.*
70512-852-14 120 mg label - 70512 852 14 120 mg label

NDC 70512-0852-14 is a medication in the form of delayed-release capsules containing 120mg dimethyl fumarate per capsule. The recommended dosage is one capsule taken orally twice a day. These capsules should be swallowed whole and stored at controlled room temperature, between 20 to 25°C, with excursions permitted to 15 to 30°C. Protect from light and dispense in the original package. This medication is prescription only and comes in a package of 14 capsules. Manufactured for Sola Pharmaceuticals in Baton Rouge, LA.*
70512-853-carton 240 mg - 70512 853 carton 240 mg

The text appears to be a medicine label. The medication is named "Swallow capsule whale" and comes in delayed-release capsules. It contains 240mg of dmaty umarte per capsule and is to be taken orally. The usual dosage is one to two capsules per day. It is available only with a prescription and comes in a package that includes dosage instructions. The medication should be stored at room temperature and protected from light. The manufacturer is Sol Pharmaceuticals. It is not clear what the medication is used for.*
70512-853-label 240 mg - 70853 853 60 label 240 mg

This is a medication called dimethyl fumarate, contained in delayed-release capsules. Each capsule contains 240 mg of the medication. The usual dosage is to take one capsule by mouth twice a day. The medication should be stored at 20-25°C (68-77°F) and protected from light. The medication is only available by prescription and the package insert should be consulted for dosage and administration information. These capsules are manufactured by Sola Pharmaceuticals, and the medication comes in a package of 60 capsules.*
figure - dimethyl fumarate dr capsules 2

This is a graph showing the percentage of subjects with confirmed progression of disability over time on a study. The X-axis represents the time on study in weeks and the Y-axis represents the percentage of subjects. The study compares the effectiveness of two treatments: Placabo (md08) and Dimetyt fumaratedeayedesense. The note on the graph indicates that confirmed progression of disability is defined as at least a 10 point increase on the EDSS from a baseline EDSS of >=10 confirmed for 12 weeks or at least a 15 point increase on the EDSS from a baseline EDSS of 0 confirmed for 12 weeks. There is additional information contained in the percentages, but without more context, it is not clear what the percentages represent.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.