Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- Structural Formula - 41d7651e b91a 4118 8719 bf2f663d82a2 01
- Figure 1 - 41d7651e b91a 4118 8719 bf2f663d82a2 02
- Figure 2 - 41d7651e b91a 4118 8719 bf2f663d82a2 03
- Figure 3 - 41d7651e b91a 4118 8719 bf2f663d82a2 04
- Figure 4 - 41d7651e b91a 4118 8719 bf2f663d82a2 05
- Figure 5 - 41d7651e b91a 4118 8719 bf2f663d82a2 06
- Figure 6 - 41d7651e b91a 4118 8719 bf2f663d82a2 07
- Figure 7 - 41d7651e b91a 4118 8719 bf2f663d82a2 08
- Figure 8 - 41d7651e b91a 4118 8719 bf2f663d82a2 09
- Figure 9 - 41d7651e b91a 4118 8719 bf2f663d82a2 10
- Figure 10 - 41d7651e b91a 4118 8719 bf2f663d82a2 11
- Donepezil HCl 5mg 70518 0452 00
Product Label Images
The following 12 images provide visual information about the product associated with Donepezil Hydrochloride NDC 70518-0452 by Remedyrepack Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - 41d7651e b91a 4118 8719 bf2f663d82a2 03

This appears to be a graph that shows the change in ADAS-cog score for a placebo group and two different treatment groups. The x-axis shows the different points in time, while the y-axis shows the percentage of patients who experienced a change in their ADAS-cog score. There are two treatment groups indicated: a group taking 10 mg/day of Donepezil hydrochloride and a group taking 5 mg/day of the same drug. The graph seems to indicate that both treatment groups had a higher percentage of patients with reduced ADAS-cog scores compared to the placebo group. However, without additional context, it is uncertain what the significance of this information is.*
Figure 3 - 41d7651e b91a 4118 8719 bf2f663d82a2 04

This appears to be a table showing the results of a study on the effectiveness of Donepezil hydrochloride and a placebo on patients, with a focus on their change in condition according to the CIBIC-plus Rating. Without more information, it is difficult to determine the context, purpose, or conclusion of the study.*
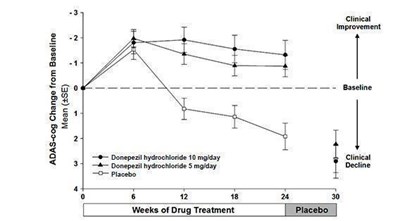
Figure 4 - 41d7651e b91a 4118 8719 bf2f663d82a2 05

The text describes a chart showing ADAS-cog change from baseline for patients receiving different treatments. The mean is represented by "+5E" and three lines indicate the treatments: donepezil hydrochloride at 10 mg/day, donepezil hydrochloride at 5 mg/day, and placebo. The chart shows results for weeks of drug treatment and suggests some clinical improvement.*
Figure 5 - 41d7651e b91a 4118 8719 bf2f663d82a2 06

The text describes a table or chart showing the percentage of patients and their ADAS-cog scores for three different treatment groups: 7 points, 4 points, and no change. The treatment groups include Donepezil hydrochloride at 10 mg/day, Donepezil hydrochloride at 5 mg/day, and a placebo. The table indicates that there was a small improvement for the 10 mg/day group (5%) and a slight worsening for the placebo group (8%), but it is unclear what the numbers 7 and 4 represent. Lastly, there is a mention of a "Chsnne foms Buselin" but it is not clear what this means.*
Figure 6 - 41d7651e b91a 4118 8719 bf2f663d82a2 07

This appears to be a table or chart related to medication dosages (Aricept 10mg/day and Aricept 5mg/day) and patient improvement ratings (Markedly Improved, Moderately Improved, Minimally Improved, No Change, Minimally Worse, Moderately Worse, Markedly Worse) as measured by the CIBIC-plus rating system. Unfortunately, without more context or information about the chart's purpose, audience or source, the description cannot be more specific.*
Figure 8 - 41d7651e b91a 4118 8719 bf2f663d82a2 09

This is a medication prescription for "Donepezil Hydrochloride" with a recommended dosage of 10mg per day. The text also includes the word "placebo". There are some illegible characters and symbols present in the text. Hence, complete understanding from the given text is not possible, and there is no significant information available.*
Donepezil HCl 5mg 70518 0452 00

Donepezil HCI is a medication that comes in tablet form with a strength of 5 mg. The package contains 90 tablets and is only available through prescription. The medication is manufactured by Sclce, Cranbury, NJ 08512, and repackaged by RemedyRepack Inc., Indiana, PA 15701. The drug should be kept out of reach of children and stored between 20-25°C (58-77°F) with short excursions permitted to 15-30°C (59-86°F) [See USP]. For further details on how to use it, please refer to the package insert accompanying the drug. The lot number and expiration date are also mentioned in the package.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.