Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 70518-0478 by Remedyrepack Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Venlafaxine HCl 37.5mg 70518 0478 00

This is a description of Venlafaxine HCI medication produced by Zydus Pharm, Pennington, NJ 08534. It comes in a capsule form and has a quantity of 30. The package insert contains the directions for use, and it should be kept out of reach of children. The medication should be stored in a temperature range of 20-25°C (58-77°F) with permitted excursions to 15-30°C (59-86°F) and has been repackaged by RemedyRepack Inc, Indiana, PA 15701. The lot number, source NDC, and expiration date for the medication are available.*
Venlafaxine HCl 37.5mg 70518 0478 01

This is a medication description for Venlafaxine HCI Extended-Release 37.5 mg capsules. It comes in a quantity of 90 and requires a prescription. The NDC number is 70518-0478-01 and the lot number, as well as the manufacturer, Zydus Pharm, is provided. The medication should be kept out of reach of children and stored at a temperature between 20-25°C (58-77°F), with excursions allowed up to 15-30°C (59-86°F). The provided directions for use are in the package insert. It has been repackaged by RemedyRepack Inc. in Indiana, PA.*
e5d724aa bc22 4503 b0e0 87b3b1d4a77e 01
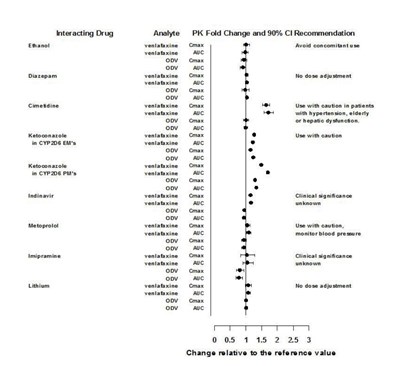
This is a list of interacting drugs with Ethanol, Diazepam, Cimetdine, and Ketoconazole in CYP2D6 EM's and PM's. It also includes recommended PK fold change and 90% C max, AUC, and clinical significance for certain drugs. It is unclear what the purpose of this list is for.*
e5d724aa bc22 4503 b0e0 87b3b1d4a77e 02
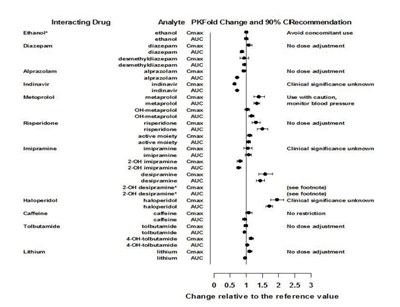
This appears to be a table listing different drugs and their interactions with each other. The listed drugs include Diazepam, Alprazolam, Indinavir, Metoprolol, Risperidone, Imipramine, Haloperidol, Caffeine, and Lithium. The table also shows PKFold change and 90% CRecommendation for each analyte, as well as Cmax and Auc values.*
e5d724aa bc22 4503 b0e0 87b3b1d4a77e 03
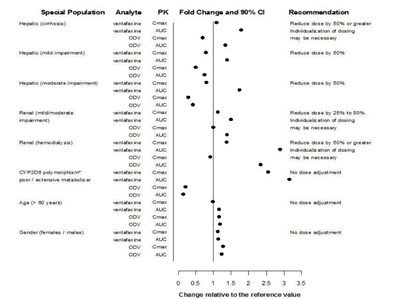
This is a report about the effects of specific populations and various impairments on the dosage requirements of the drug analyte venlafaxine. The populations studied include those with hepatic and renal impairments, CYP2D8 polymorphism, and advanced age or gender. For those with impairments or polymorphism, a reduction in dosage is recommended, while no adjustment is needed for the other populations. The report provides fold changes and 90% confidence intervals for the required dose changes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.