Product Images Clopidogrel
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Clopidogrel NDC 70518-1510 by Remedyrepack Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Remedy_Label - Remedy Label

Clopidogrel is a medication that comes in tablet form with each tablet containing 75mg of the active ingredient. The package comes with 30 tablets and is RX only. The medication is manufactured by Accord Healthcare, Inc., Durham, NC, and repackaged by RemedyRepack Inc., Indiana, PA. The National Drug Code (NDC) is 70518-1510-00, and the Lot number and expiry are not available. Store the medication at the recommended temperature range of 20-25°C (88-77°F), and keep it away from children. The package insert should be followed for directions for use.*
Figure - clopidogrel fig1

This is a chart showing the cumulative event rate of fatal and non-fatal vascular events for patients taking Aspirin versus Clopidogrel Bisulfate over a follow-up period of 36 months. The chart indicates that the event rate for Aspirin is slightly higher than for Clopidogrel Bisulfate, with a statistically significant difference noted at 0.045% (p-value).*
Figure - clopidogrel fig2

The text shows a chart titled "Cumulative Event Rate (%) Cardiovascular death, myocardial infarction, stroke" comparing results between a placebo (with aspirin) and clopidogrel bisulfate (with aspirin) over a follow-up period of 12 months. The chart shows the placebo group has a median event rate of 16% while clopidogrel bisulfate group has a lower median event rate of less than 1.2%. It also mentions that other standard therapies were used as appropriate. Lastly, there is a significance value of P=0.00009.*
Figure - clopidogrel fig4

This text provides statistical data for a clinical trial of a medication called Clopidogrel. The statistic shows that the medication caused a 7% reduction in death rates compared to a placebo, with 1845 deaths occurring in the placebo group and 1726 deaths occurring in the Clopidogrel group. The reduction has a p-value of 0.03. The data is tracked up to 28 days since randomization.*
Figure - clopidogrel fig5

This text appears to be a portion of a clinical trial result that discusses the incidence of death, re-infarction or stroke before first discharge among two groups: one treated with placebo and the other treated with Clopidogrel. The study shows that Clopidogrel has a 9% proportional risk reduction for these incidents (with a p value of 0.002), compared to the placebo. The text also mentions randomization, up to 28 days.*
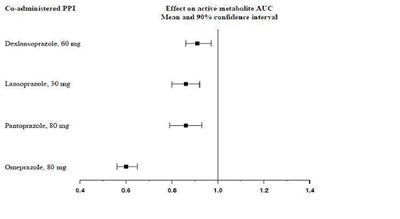
Figure - clopidogrel fig9

This appears to be a table showing the results of a study comparing the effectiveness of Clopidogrel and Aspirin in patients with stroke or PAD (peripheral arterial disease). The table includes the number of patients in each group, as well as the percentage of patients receiving each medication. The table also includes results from different years of the study, and a hazard ratio with a confidence interval.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.