Product Images Ipratropium Bromide And Albuterol Sulfate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Ipratropium Bromide And Albuterol Sulfate NDC 70518-2250 by Remedyrepack Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Ipratropium Bromide Albuterol Sulfate 0.5mg 3mg 3mL 70518 2250 01

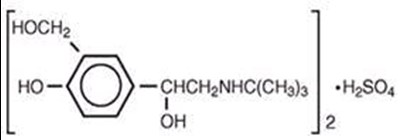
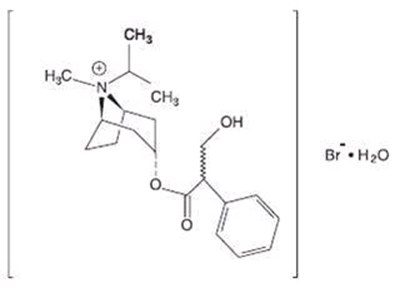
This is a description of medication called Ipratropium which has a Bromide/Albuterol content of 2.5mg albuterol base. It comes in inhalation solution form with a quantity of 90. The medication is manufactured by Nephron Pharma Corp in Orlando, FL. The package insert holds the directions for use and it should be kept out of children's reach. The medication should be stored at 20-26°C (63-77°F), with excursions permitted to 15-30°C (59-86°F), and is designated for pharmacy prescription only.*
Remedy_Label - Remedy Label

This is a medication called Ipratropium Bromide/Albuterol Sulfate. It comes in the form of inhalation solution with a quantity of 180. The LOT number is equivalent to 2.5mg of albuterol base. The manufacturing company of this medication is Nephron Pharma Corp, located in Orlando, FL. The storage temperature for this medication is between 20-26°C. The package insert should be consulted for directions of use. Keep this medication out of children’s reach.*
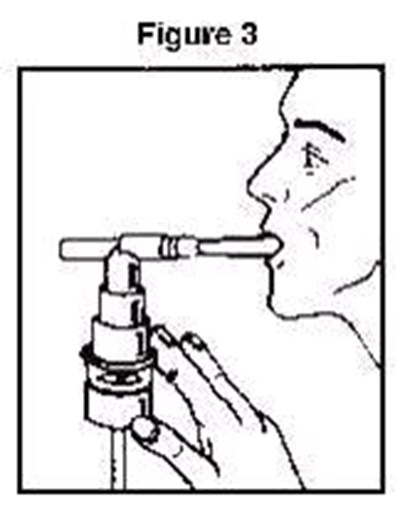
b5387e98-figure-03 - b5387e98 figure 03
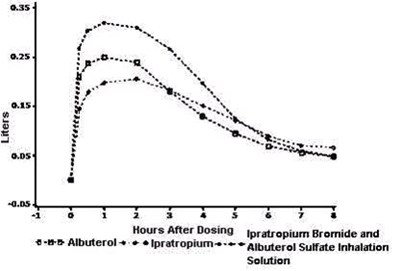
The text appears to be a description of a medication called "mimutoral Sulfote Inhalation Solution", which is a combination of Albuterol, Ipratropium and some unidentified chemical represented by the symbol "+o+". The medication seems to be used for inhalation purposes and could potentially be used for treating respiratory conditions. The text also includes some numerical values (.15, 2§ and o081) which appear to be unrelated to the medication.*
b5387e98-figure-04 - b5387e98 figure 04
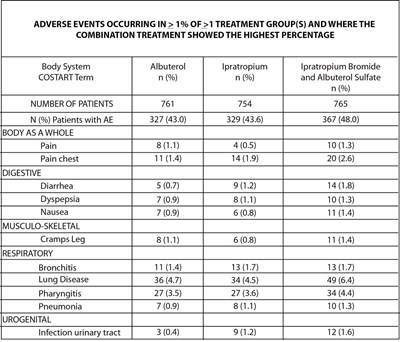
This document presents a table with adverse events occurring in different treatment groups (Albuterol, Ipratropium, and Ipratropium bromide) and where the combination treatment showed the highest percentage. The adverse events are grouped by body system (e.g., Respiratory, Digestive, etc.) and some of the events reported include Bronchitis, Lung Disease, Pneumonia, Pain, Diarrhea, and Infection of the urinary tract.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.