Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
- Remedy_Label - Remedy Label
- Figure 1 - pregabalin fig01
- Figure 2 - pregabalin fig02
- Figure 3 - pregabalin fig03
- Figure 4 - pregabalin fig04
- Figure 5 - pregabalin fig05
- Figure 6 - pregabalin fig06
- Figure 7 - pregabalin fig07
- Figure 9 - pregabalin fig08
- Figure 10 - pregabalin fig09
- Figure 11 - pregabalin fig10
- Figure 12 - pregabalin fig11
- Figure 13 - pregabalin fig13
- Cockcroft and Gault equation - pregabalin formula
- Chemical Structure - pregabalin str
Product Label Images
The following 15 images provide visual information about the product associated with Pregabalin NDC 70518-2257 by Remedyrepack Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Remedy_Label - Remedy Label

Pregabalin is an oral medication available in the form of capsules. Each capsule contains 150 mg of the active ingredient. This package contains a total of 30 capsules. The medication is labeled for prescription use only. It is identified by the National Drug Code (NDC) number 70518-2257-00. The expiration date and lot number of the product are not available. The original source NDC is 50228-0354-90. The medication is manufactured by ScieGen Pharmaceuticals, Inc. located in Hauppaugs, NY 11788. It is important to keep this medication out of the reach of children. The directions for use can be found in the package insert. The recommended storage temperature is between 20-25°C (58-77°F), with excursions allowed up to 15-30°C (59-86°F) as per USP guidelines. The medication has been repackaged by RemedyRepack Inc. located in Indiana, PA 15701, and their contact number is 724.465.8762.*
Figure 5 - pregabalin fig05
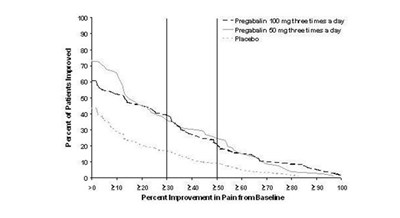
This text appears to be a record of a medication regimen for two drugs, Fregabalin and Pregataiin, along with a placebo. The dosage for Fregabalin is 100mg three times a day, and the dosage for Pregataiin is 80mg three times a day. It also mentions a percentage of patients improved, but the exact value is not clear. Additionally, there is a representation of percentages indicating pain improvement from a baseline measurement.*
Figure 12 - pregabalin fig11
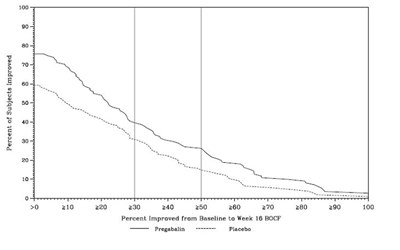
The text provided indicates the percentage of subjects who showed improvement over a certain period. The values mentioned are 100%, 210%, 220%, 230%, 240%, 250%, 260%, 270%, 280%, and 200%. The text also mentions "Pregabalin" and "Placebo" which may indicate different treatment groups. However, the context and additional information are not available to provide a more specific description.*
Figure 13 - pregabalin fig13

Responder Rate (%): This text provides information about the responder rate expressed in percentage. The responder rate starts at 60% and gradually decreases to 50%. After that, the values are not readable due to the error. The next readable values indicate responder rates of 40.6%, 29.1%, and 22.6%. The following text mentions "Placebo 2.5 mglkglday 10 mg/kglday" followed by numbers 93, 103, and 9%. The specific details about the intervention or treatment are not clear, but it appears to involve different dosages.*
Cockcroft and Gault equation - pregabalin formula
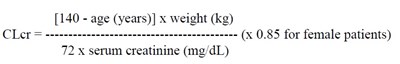
The given text appears to be a formula for calculating CLCT (Creatinine Clearance) using the patient's age, weight, and serum creatinine level. However, some characters in the text are not readable due to errors, resulting in "mmmmmmmmm" and "e e". Therefore, the description is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.









