Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
- Remedy_Label - Remedy Label
- image fig1 effectofotherdrug arip
- image fig2 effectofotherdrug dehydroarip
- image fig3 effectofarip otherdrug
- image fig4 effectofintrinsicfactor arip
- image fig5 effectofintrinsicfactor dehydroarip
- image fig6 schizophreniastudy 5
- image fig7 bipolarstudy 7
- image fig8 bipolarstudy 8
- image fig9 tourettesdisorderstudy1
Product Label Images
The following 10 images provide visual information about the product associated with Aripiprazole NDC 70518-2536 by Remedyrepack Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Remedy_Label - Remedy Label

Aripiprazole is a medication that comes in 5mg tablets. The National Drug Code for this medication is 70518-2536-00 and it expires on an unknown date. The manufacturer of the medication is Torrent Pharma Inc, NJ, and the medication is packaged with a quantity of 30 tablets per package. It is important to keep this and all other medication out of reach of children. The recommended storage temperature is between 15-30°C (59-86°F). The use of this medication should be done in accordance with the instructions found in the package insert. This medication is available by prescription only and it was repackaged by RemedyRepack Inc, PA.*
image fig1 effectofotherdrug arip
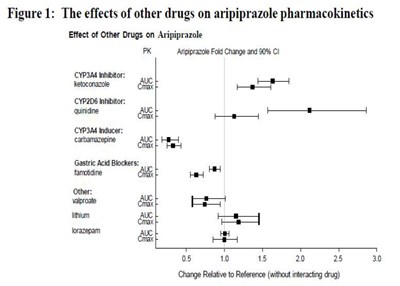
This is a chart showing the effects of different drugs on the pharmacokinetics of Aripiprazole, a medication often used to treat schizophrenia and bipolar disorder. The chart displays Aripiprazole's fold change and 90% confidence interval when interacting with specific drugs like ketoconazole, quinidine, carbamazepine, famidine, valproate, lithium, and lorazepam. The fold change is relative to the reference without the interacting drug.*
image fig3 effectofarip otherdrug

This is a figure displaying the effects of aripiprazole on the pharmacokinetics of several other drugs, including lamotrigine, lithium, omeprazole, lorazepam, and escitalopram. The fold changes and 50% confidence intervals are shown on the y-axis, and the drugs are listed on the x-axis. The graph shows the relative change to the reference (without the interacting drug) from 0 to 30 on the x-axis.*
image fig4 effectofintrinsicfactor arip

The image is a figure that shows the effects of intrinsic factors on the pharmacokinetics of aripiprazole, an antipsychotic medication. The figure includes comparisons of different special populations, such as poor versus extensive metabolizers, males versus females, and individuals of different ages. Additionally, the figure compares the pharmacokinetics of aripiprazole in individuals with varying degrees of hepatic and renal impairment. The information is summarized in fold changes and 90% confidence intervals for AUC and Cmax values.*
image fig5 effectofintrinsicfactor dehydroarip

The image shows the effects of different intrinsic factors on the pharmacokinetics of dehydro-aripiprazole, presented in fold change and S0% CI. These factors include CYP2D6 metabolizer status, gender, age, hepatic impairment, and renal impairment. The graph displays the changes relative to the reference values for each factor.*
image fig6 schizophreniastudy 5

This is a graph representing the cumulative proportion of relapse in patients with schizophrenia study. The graph shows the comparison between aripiprazole and placebo. The number of subjects at risk is also shown. The x-axis shows the total number of days from randomization.*
image fig7 bipolarstudy 7
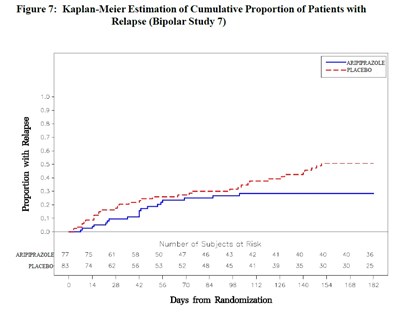
The figure shows the Kaplan-Meier estimation of the cumulative proportion of patients with relapse in Bipolar Study 7. The graph displays two groups, Aripiprazole, and Placebo. The number of subjects at risk is displayed below the graph. The X-axis displays the number of days from randomization, starting with 0 days and ending with 182 days.*
image fig9 tourettesdisorderstudy1
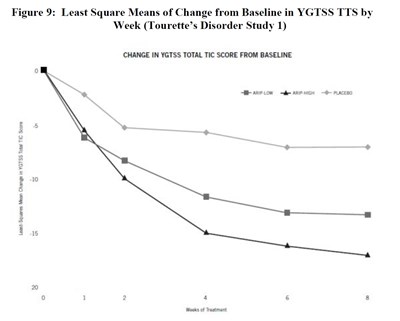
This is a graph showing the least square means of change from baseline in YGTSS TTS by week in a Tourette's Disorder Study. The text also includes the change in YGTSS total tic score from baseline and a table with the weeks of treatment and least square means change in YGTS.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

